RETRACTED: KRAS Activation in Gastric Adenocarcinoma Stimulates Epithelial-to-Mesenchymal Transition to Cancer Stem-Like Cells and Promotes Metastasis
- PMID: 31217166
- PMCID: PMC6726517
- DOI: 10.1158/1541-7786.MCR-19-0077
RETRACTED: KRAS Activation in Gastric Adenocarcinoma Stimulates Epithelial-to-Mesenchymal Transition to Cancer Stem-Like Cells and Promotes Metastasis
Retraction in
-
Retraction: KRAS Activation in Gastric Adenocarcinoma Stimulates Epithelial-to-Mesenchymal Transition to Cancer Stem-Like Cells and Promotes Metastasis.Mol Cancer Res. 2024 Nov 1;22(11):1066. doi: 10.1158/1541-7786.MCR-24-0858. Mol Cancer Res. 2024. PMID: 39482978 Free PMC article. No abstract available.
Abstract
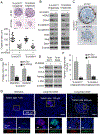
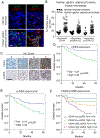
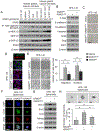
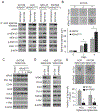
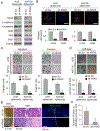
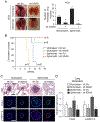
Our previous work showed that in a mouse model of gastric adenocarcinoma with loss of p53 and Cdh1 that adding oncogenic Kras (a.k.a. Tcon mice) accelerates tumorigenesis and metastasis. Here, we sought to examine KRAS activation in epithelial-to-mesenchymal transition (EMT) and generation of cancer stem-like cells (CSC). Transduction of nontransformed HFE-145 gastric epithelial cells with oncogenic KRASG12V significantly decreased expression of the epithelial marker E-cadherin, increased expression of the mesenchymal marker vimentin and the EMT transcription factor Slug, and increased migration and invasion by 15- to 17-fold. KRASG12V also increased expression of self-renewal proteins such as Sox2 and increased spheroid formation by 2.6-fold. In tumor-derived organoids from Tcon mice, Kras knockdown decreased spheroid formation, expression of EMT-related proteins, migration, and invasion; similar effects, as well as reversal of chemoresistance, were observed following KRAS knockdown or MEK inhibition in patient tumor-derived gastric adenocarcinoma cell lines (AGS and KATOIII). KRAS inhibition in gastric adenocarcinoma spheroid cells led to reduced AGS flank xenograft growth, loss of the infiltrative tumor border, fewer lung metastases, and increased survival. In a tissue microarray of human gastric adenocarcinomas from 115 patients, high tumor levels of CD44 (a marker of CSCs) and KRAS activation were independent predictors of worse overall survival. In conclusion, KRAS activation in gastric adenocarcinoma cells stimulates EMT and transition to CSCs, thus promoting metastasis. IMPLICATIONS: This study provides rationale for examining inhibitors of KRAS to block metastasis and reverse chemotherapy resistance in gastric adenocarcinoma patients.
©2019 American Association for Cancer Research.
Conflict of interest statement
Conflict of interest: All authors have no conflicts of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

