In silico simulation of a clinical trial with anti-CTLA-4 and anti-PD-L1 immunotherapies in metastatic breast cancer using a systems pharmacology model
- PMID: 31218069
- PMCID: PMC6549962
- DOI: 10.1098/rsos.190366
In silico simulation of a clinical trial with anti-CTLA-4 and anti-PD-L1 immunotherapies in metastatic breast cancer using a systems pharmacology model
Abstract
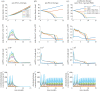
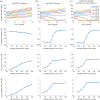
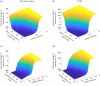
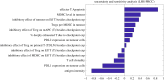
The low response rate of immune checkpoint blockade in breast cancer has highlighted the need for predictive biomarkers to identify responders. While a number of clinical trials are ongoing, testing all possible combinations is not feasible. In this study, a quantitative systems pharmacology model is built to integrate immune-cancer cell interactions in patients with breast cancer, including central, peripheral, tumour-draining lymph node (TDLN) and tumour compartments. The model can describe the immune suppression and evasion in both TDLN and the tumour microenvironment due to checkpoint expression, and mimic the tumour response to checkpoint blockade therapy. We investigate the relationship between the tumour response to checkpoint blockade therapy and composite tumour burden, PD-L1 expression and antigen intensity, including their individual and combined effects on the immune system, using model-based simulations. The proposed model demonstrates the potential to make predictions of tumour response of individual patients given sufficient clinical measurements, and provides a platform that can be further adapted to other types of immunotherapy and their combination with molecular-targeted therapies. The patient predictions demonstrate how this systems pharmacology model can be used to individualize immunotherapy treatments. When appropriately validated, these approaches may contribute to optimization of breast cancer treatment.
Keywords: computational biology; computational model; immune checkpoint inhibitor; immuno-oncology; systems biology.
Conflict of interest statement
C.A.S.-M. receives research support from Pfizer and MedImmune and serves on the advisory board for Polyphor. I.H.B., P.V., B.W., R.N. and L.R. were employees of MedImmune. A.S.P. receives research support from MedImmune. Other authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures






Similar articles
-
Quantitative systems pharmacology model predictions for efficacy of atezolizumab and nab-paclitaxel in triple-negative breast cancer.J Immunother Cancer. 2021 Feb;9(2):e002100. doi: 10.1136/jitc-2020-002100. J Immunother Cancer. 2021. PMID: 33579739 Free PMC article.
-
Conducting a Virtual Clinical Trial in HER2-Negative Breast Cancer Using a Quantitative Systems Pharmacology Model With an Epigenetic Modulator and Immune Checkpoint Inhibitors.Front Bioeng Biotechnol. 2020 Feb 25;8:141. doi: 10.3389/fbioe.2020.00141. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 32158754 Free PMC article.
-
The Next Immune-Checkpoint Inhibitors: PD-1/PD-L1 Blockade in Melanoma.Clin Ther. 2015 Apr 1;37(4):764-82. doi: 10.1016/j.clinthera.2015.02.018. Epub 2015 Mar 29. Clin Ther. 2015. PMID: 25823918 Free PMC article. Review.
-
Radiation and PD-(L)1 treatment combinations: immune response and dose optimization via a predictive systems model.J Immunother Cancer. 2018 Feb 27;6(1):17. doi: 10.1186/s40425-018-0327-9. J Immunother Cancer. 2018. PMID: 29486799 Free PMC article.
-
Targeting immune checkpoints in breast cancer: an update of early results.ESMO Open. 2017 Nov 14;2(5):e000255. doi: 10.1136/esmoopen-2017-000255. eCollection 2017. ESMO Open. 2017. PMID: 29177095 Free PMC article. Review.
Cited by
-
Dynamics of tumor-associated macrophages in a quantitative systems pharmacology model of immunotherapy in triple-negative breast cancer.iScience. 2022 Jun 30;25(8):104702. doi: 10.1016/j.isci.2022.104702. eCollection 2022 Aug 19. iScience. 2022. PMID: 35856032 Free PMC article.
-
A Spatial Quantitative Systems Pharmacology Platform spQSP-IO for Simulations of Tumor-Immune Interactions and Effects of Checkpoint Inhibitor Immunotherapy.Cancers (Basel). 2021 Jul 26;13(15):3751. doi: 10.3390/cancers13153751. Cancers (Basel). 2021. PMID: 34359653 Free PMC article.
-
Endogenous pAKT activity is associated with response to AKT inhibition alone and in combination with immune checkpoint inhibition in murine models of TNBC.Cancer Lett. 2024 Apr 1;586:216681. doi: 10.1016/j.canlet.2024.216681. Epub 2024 Feb 3. Cancer Lett. 2024. PMID: 38311054 Free PMC article.
-
Toward Systems Biomarkers of Response to Immune Checkpoint Blockers.Front Oncol. 2020 Jun 24;10:1027. doi: 10.3389/fonc.2020.01027. eCollection 2020. Front Oncol. 2020. PMID: 32670886 Free PMC article. Review.
-
In silico dynamics of COVID-19 phenotypes for optimizing clinical management.Proc Natl Acad Sci U S A. 2021 Jan 19;118(3):e2021642118. doi: 10.1073/pnas.2021642118. Proc Natl Acad Sci U S A. 2021. PMID: 33402434 Free PMC article.
References
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

