Depressive Symptoms Assessed Near the End of Pregnancy Predict Differential Response to Postpartum Smoking Relapse Prevention Intervention
- PMID: 31219152
- PMCID: PMC7170725
- DOI: 10.1093/abm/kaz026
Depressive Symptoms Assessed Near the End of Pregnancy Predict Differential Response to Postpartum Smoking Relapse Prevention Intervention
Abstract
Background: Depressive symptoms are prevalent during pregnancy and the postpartum period and affect risk for smoking relapse. Whether and how depression affects response to postpartum interventions designed to sustain smoking abstinence is unknown.
Purpose: We examined end-of-pregnancy depressive symptoms as a moderator of response to two postpartum-adapted smoking relapse prevention interventions.
Methods: Women (N = 300) who quit smoking during pregnancy were randomized to receive either a postpartum intervention focused on psychosocial factors linked to postpartum smoking (Strategies to Avoid Returning to Smoking [STARTS]) or an attention-controlled comparison intervention (SUPPORT). Women completed the Edinburgh Postnatal Depression Scale at the end of pregnancy. Smoking status was biochemically assessed at the end of pregnancy and at 12, 24, and 52 weeks postpartum.
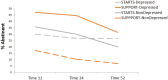
Results: End-of-pregnancy depressive symptoms moderated response to postpartum smoking relapse prevention interventions (χ2 = 10.18, p = .001). After controlling for variables previously linked to postpartum smoking relapse, women with clinically significant end-of-pregnancy depressive symptoms (20%) were more likely to sustain abstinence through 52 weeks postpartum if they received STARTS. In contrast, women with few end-of-pregnancy depressive symptoms were more likely to sustain abstinence through 52 weeks postpartum if they received SUPPORT. Changes in the psychosocial factors addressed in the STARTS intervention did not mediate this moderation effect.
Conclusion: Assessment of end-of-pregnancy depressive symptoms may help determine success following postpartum smoking relapse prevention interventions. Women with elevated end-of-pregnancy depressive symptoms benefited from postpartum relapse prevention intervention tailored to their psychosocial needs, while those with few symptoms were more successful in postpartum intervention that used standard behavioral components.
Clinical trial registration: NCT00757068.
Keywords: Depressive symptoms; Postpartum; Relapse prevention; Smoking cessation.
© Society of Behavioral Medicine 2019. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
Similar articles
-
Do Psychiatric Disorders or Measures of Distress Moderate Response to Postpartum Relapse Prevention Interventions?Nicotine Tob Res. 2017 May 1;19(5):615-622. doi: 10.1093/ntr/ntw385. Nicotine Tob Res. 2017. PMID: 28403471 Free PMC article.
-
Preventing Postpartum Smoking Relapse: A Randomized Clinical Trial.JAMA Intern Med. 2016 Apr;176(4):443-52. doi: 10.1001/jamainternmed.2016.0248. JAMA Intern Med. 2016. PMID: 26998789 Free PMC article. Clinical Trial.
-
Strategies to Avoid Returning to Smoking (STARTS): a randomized controlled trial of postpartum smoking relapse prevention interventions.Contemp Clin Trials. 2013 Nov;36(2):565-73. doi: 10.1016/j.cct.2013.10.002. Epub 2013 Oct 17. Contemp Clin Trials. 2013. PMID: 24140455 Free PMC article. Clinical Trial.
-
Smoking cessation in pregnancy: a review of postpartum relapse prevention strategies.J Am Board Fam Pract. 2004 Jul-Aug;17(4):264-75. doi: 10.3122/jabfm.17.4.264. J Am Board Fam Pract. 2004. PMID: 15243014 Review.
-
Maintenance of smoking cessation in the postpartum period: which interventions work best in the long-term?Matern Child Health J. 2014 Apr;18(3):714-28. doi: 10.1007/s10995-013-1298-6. Matern Child Health J. 2014. PMID: 23812798 Review.
References
-
- Hannöver W, Thyrian JR, Röske K, et al. . Smoking cessation and relapse prevention for postpartum women: Results from a randomized controlled trial at 6, 12, 18 and 24 months. Addict Behav. 2009;34:1–8. - PubMed

