Redox Chemistry in the Genome: Emergence of the [4Fe4S] Cofactor in Repair and Replication
- PMID: 31220976
- PMCID: PMC6590699
- DOI: 10.1146/annurev-biochem-013118-110644
Redox Chemistry in the Genome: Emergence of the [4Fe4S] Cofactor in Repair and Replication
Abstract
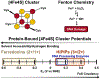
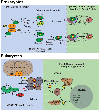
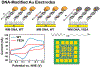
Many DNA-processing enzymes have been shown to contain a [4Fe4S] cluster, a common redox cofactor in biology. Using DNA electrochemistry, we find that binding of the DNA polyanion promotes a negative shift in [4Fe4S] cluster potential, which corresponds thermodynamically to a ∼500-fold increase in DNA-binding affinity for the oxidized [4Fe4S]3+ cluster versus the reduced [4Fe4S]2+ cluster. This redox switch can be activated from a distance using DNA charge transport (DNA CT) chemistry. DNA-processing proteins containing the [4Fe4S] cluster are enumerated, with possible roles for the redox switch highlighted. A model is described where repair proteins may signal one another using DNA-mediated charge transport as a first step in their search for lesions. The redox switch in eukaryotic DNA primases appears to regulate polymerase handoff, and in DNA polymerase δ, the redox switch provides a means to modulate replication in response to oxidative stress. We thus describe redox signaling interactions of DNA-processing [4Fe4S] enzymes, as well as the most interesting potential players to consider in delineating new DNA-mediated redox signaling networks.
Keywords: DNA charge transport; DNA polymerase; DNA primase; base excision repair; iron–sulfur clusters; oxidative stress; redox signaling.
Figures


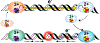




References
-
- Beinert H, Holm RH, Munck E. 1997. Iron-sulfur clusters: Nature’s modular, multipurpose structures. Science 277:653–59 - PubMed
-
- Rees DC, Howard JB. 2003. The interface between the biological and inorganic worlds: Iron-sulfur metalloclusters. Science 300:929–31 - PubMed
-
- Meyer J 2008. Iron-sulfur protein folds, iron-sulfur chemistry, and evolution. J. Biol. Inorg. Chem 13:157–70 - PubMed
-
- Dey A, Jenney FE, Adams MWW, Babini E, Takahashi Y, et al. 2007. Solvent tuning of electrochemical potentials in the active sites of HiPIP versus ferredoxin. Science 318:1464–68 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

