Predictors of disease worsening defined by progression of organ damage in diffuse systemic sclerosis: a European Scleroderma Trials and Research (EUSTAR) analysis
- PMID: 31227488
- PMCID: PMC6788922
- DOI: 10.1136/annrheumdis-2019-215145
Predictors of disease worsening defined by progression of organ damage in diffuse systemic sclerosis: a European Scleroderma Trials and Research (EUSTAR) analysis
Abstract
Objectives: Mortality and worsening of organ function are desirable endpoints for clinical trials in systemic sclerosis (SSc). The aim of this study was to identify factors that allow enrichment of patients with these endpoints, in a population of patients from the European Scleroderma Trials and Research group database.
Methods: Inclusion criteria were diagnosis of diffuse SSc and follow-up over 12±3 months. Disease worsening/organ progression was fulfilled if any of the following events occurred: new renal crisis; decrease of lung or heart function; new echocardiography-suspected pulmonary hypertension or death. In total, 42 clinical parameters were chosen as predictors for the analysis by using (1) imputation of missing data on the basis of multivariate imputation and (2) least absolute shrinkage and selection operator regression.
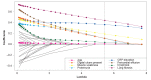
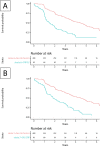
Results: Of 1451 patients meeting the inclusion criteria, 706 had complete data on outcome parameters and were included in the analysis. Of the 42 outcome predictors, eight remained in the final regression model. There was substantial evidence for a strong association between disease progression and age, active digital ulcer (DU), lung fibrosis, muscle weakness and elevated C-reactive protein (CRP) level. Active DU, CRP elevation, lung fibrosis and muscle weakness were also associated with a significantly shorter time to disease progression. A bootstrap validation step with 10 000 repetitions successfully validated the model.
Conclusions: The use of the predictive factors presented here could enable cohort enrichment with patients at risk for overall disease worsening in SSc clinical trials.
Keywords: disease worsening; mortality; predictive factors; systemic sclerosis.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY. Published by BMJ.
Conflict of interest statement
Competing interests: MOB declares no conflict of interest. OD has had consultancy relationships with Actelion, Bayer, Biogen Idec, Boehringer Ingelheim, ChemomAb, espeRare foundation, Genentech/Roche, GSK, Inventiva, Italfarmaco, Lilly, medac, MedImmune, Mitsubishi Tanabe Pharma, Pharmacyclics, Novartis, Pfizer, Sanofi, Sinoxa and UCB in the area of potential treatments of scleroderma and its complications. OD has received research funding from Actelion, Bayer, Boehringer Ingelheim, Mitsubishi Tanabe Pharma and Roche in the area of potential treatments of scleroderma and its complications. OD has a patent for mir-29 licensed for the treatment of systemic sclerosis. DK has consultancy relationships and/or has received grant/research support from Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Genentech/Roche, NIH, Pfizer, Sanofi-Aventis Pharmaceuticals, Actelion Pharmaceuticals US, ChemomAb, Corbus, Covis, Cytori, Eicos, EMD Serono, Gilead, GlaxoSmithKline, and UCB Pharma. He is a shareholder of Eicos. CPD has consultancy relationships with and/or has received speakers’ bureau fees from Actelion Pharmaceuticals US, Bayer AG, GlaxoSmithKline, CSL Behring, Merck Serono, Roche Pharmaceuticals, Genentech and Biogen IDEC Inc., Inventiva, Sanofi-Aventis Pharmaceuticals and Boehringer Ingelheim. JEP has consultancy relationships with and/or has received grant/research support from Actelion, Bayer AG, Bristol-Myers Squibb, Merck, Pfizer Inc. and Roche. MM-C has consultancy relationships and/or has received grant/research support from Pfizer, Bristol-Myers Squibb, Actelion, UCB Pharma, Bayer, ChemomAb, Genentech/Roche, Inventiva and Lilly. YA has consultancy relationships with and/or has received grant/research support from Actelion, Pharmaceuticals US, Bayer AG, Bristol-Myers Squibb, Inventiva, Medac, Pfizer Inc., Roche Pharmaceuticals, Genentech and Biogen IDEC Inc., Sanofi-Aventis Pharmaceuticals and Servier. JdOP and JC are employees of Bayer. NTG has nothing to disclose.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

