An Integrative Analysis of Tumor Proteomic and Phosphoproteomic Profiles to Examine the Relationships Between Kinase Activity and Phosphorylation
- PMID: 31227600
- PMCID: PMC6692771
- DOI: 10.1074/mcp.RA119.001540
An Integrative Analysis of Tumor Proteomic and Phosphoproteomic Profiles to Examine the Relationships Between Kinase Activity and Phosphorylation
Abstract
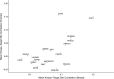
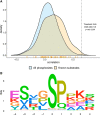
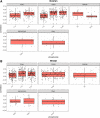
Phosphorylation of proteins is a key way cells regulate function, both at the individual protein level and at the level of signaling pathways. Kinases are responsible for phosphorylation of substrates, generally on serine, threonine, or tyrosine residues. Though particular sequence patterns can be identified that dictate whether a residue will be phosphorylated by a specific kinase, these patterns are not highly predictive of phosphorylation. The availability of large scale proteomic and phosphoproteomic data sets generated using mass-spectrometry-based approaches provides an opportunity to study the important relationship between kinase activity, substrate specificity, and phosphorylation. In this study, we analyze relationships between protein abundance and phosphopeptide abundance across more than 150 tumor samples and show that phosphorylation at specific phosphosites is not well correlated with overall kinase abundance. However, individual kinases show a clear and statistically significant difference in correlation among known phosphosite targets for that kinase and randomly selected phosphosites. We further investigate relationships between phosphorylation of known activating or inhibitory sites on kinases and phosphorylation of their target phosphosites. Combined with motif-based analysis, this approach can predict novel kinase targets and show which subsets of a kinase's target repertoire are specifically active in one condition versus another.
Keywords: Breast cancer; Cancer Biology; Computational Biology; Ovarian cancer; Phosphoproteome; Phosphorylation; iTRAQ.
© 2019 Arshad et al.
Figures




References
-
- Hanahan D., and Weinberg R. A. (2011) Hallmarks of cancer: the next generation. Cell. 144, 646–674 - PubMed
-
- Sharma K., D'Souza R. C., Tyanova S., Schaab C., Wisniewski J. R., Cox J., and Mann M. (2014) Ultradeep human phosphoproteome reveals a distinct regulatory nature of Tyr and Ser/Thr-based signaling. Cell Reports 8, 1583–1594 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

