Hexamethylenediamine-Mediated Polydopamine Film Deposition: Inhibition by Resorcinol as a Strategy for Mapping Quinone Targeting Mechanisms
- PMID: 31231635
- PMCID: PMC6560077
- DOI: 10.3389/fchem.2019.00407
Hexamethylenediamine-Mediated Polydopamine Film Deposition: Inhibition by Resorcinol as a Strategy for Mapping Quinone Targeting Mechanisms
Abstract
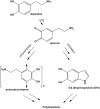
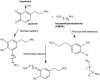
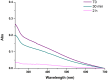
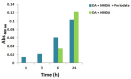
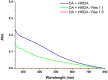
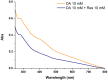
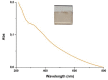
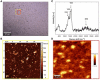
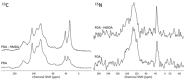
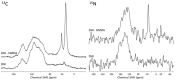
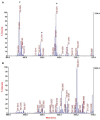
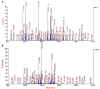
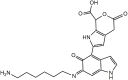
Hexamethylenediamine (HMDA) and other long chain aliphatic diamines can induce substrate-independent polymer film deposition from dopamine and several other catechols substrates at relatively low concentrations, however the mechanism of the diamine-promoted effect has remained little understood. Herein, we report data indicating that: (a) film deposition from 1 mM HMDA and dopamine is not affected by chemical oxidation with periodate but is markedly inhibited by resorcinol, which also prevents PDA film formation at 10 mM monomer concentration in the absence of HMDA; (b) N-acetylation of HMDA completely inhibits the effect on PDA film formation; (c) HMDA enables surface functionalization with 1 mM 5,6-dihydroxyindole (DHI) polymerization at pH 9.0 in a resorcinol-inhibitable manner. Structural investigation of the polymers produced from dopamine and DHI in the presence of HMDA using solid state 13C and 15N NMR and MALDI-MS suggested formation of covalent cross linked structures. It is concluded that HMDA enhances polydopamine adhesion by acting both on dopamine quinone and downstream, e.g., via covalent coupling with DHI. These results provide new insights into the mechanisms of PDA adhesion and disclose resorcinol as a new potent tool for targeting/mapping quinone intermediates and for controlling polymer growth.
Keywords: 5; 6-dihydroxyindole; coating; hexamethylenediamine; polydopamine; resorcinol.
Figures

Similar articles
-
Reaction-Based, Fluorescent Film Deposition from Dopamine and a Diamine-Tethered, Bis-Resorcinol Coupler.Int J Mol Sci. 2019 Sep 13;20(18):4532. doi: 10.3390/ijms20184532. Int J Mol Sci. 2019. PMID: 31540228 Free PMC article.
-
The Chemistry of Polydopamine Film Formation: The Amine-Quinone Interplay.Biomimetics (Basel). 2018 Sep 13;3(3):26. doi: 10.3390/biomimetics3030026. Biomimetics (Basel). 2018. PMID: 31105248 Free PMC article.
-
Non-covalent small molecule partnership for redox-active films: Beyond polydopamine technology.J Colloid Interface Sci. 2022 Oct 15;624:400-410. doi: 10.1016/j.jcis.2022.05.123. Epub 2022 May 22. J Colloid Interface Sci. 2022. PMID: 35671617
-
Structural Basis of Polydopamine Film Formation: Probing 5,6-Dihydroxyindole-Based Eumelanin Type Units and the Porphyrin Issue.ACS Appl Mater Interfaces. 2018 Mar 7;10(9):7670-7680. doi: 10.1021/acsami.7b09662. Epub 2017 Sep 29. ACS Appl Mater Interfaces. 2018. PMID: 28937213
-
Toxicity of hexamethylenediamine.Drug Chem Toxicol. 2005;28(1):15-33. doi: 10.1081/dct-39681. Drug Chem Toxicol. 2005. PMID: 15720033 Review.
Cited by
-
A Clean and Tunable Mussel-Inspired Coating Technology by Enzymatic Deposition of Pseudo-Polydopamine (ψ-PDA) Thin Films from Tyramine.Int J Mol Sci. 2020 Jul 10;21(14):4873. doi: 10.3390/ijms21144873. Int J Mol Sci. 2020. PMID: 32664213 Free PMC article.
-
Reaction-Based, Fluorescent Film Deposition from Dopamine and a Diamine-Tethered, Bis-Resorcinol Coupler.Int J Mol Sci. 2019 Sep 13;20(18):4532. doi: 10.3390/ijms20184532. Int J Mol Sci. 2019. PMID: 31540228 Free PMC article.
-
Site-specific fabrication of a melanin-like pigment through spatially confined progressive assembly on an initiator-loaded template.Nat Commun. 2023 Jun 10;14(1):3432. doi: 10.1038/s41467-023-38622-2. Nat Commun. 2023. PMID: 37301846 Free PMC article.
-
Deposition of Antioxidant and Cytocompatible Caffeic Acid-Based Thin Films onto Ti6Al4V Alloys through Hexamethylenediamine-Mediated Crosslinking.ACS Appl Mater Interfaces. 2023 Jun 21;15(24):29618-29635. doi: 10.1021/acsami.3c05564. Epub 2023 Jun 8. ACS Appl Mater Interfaces. 2023. PMID: 37289140 Free PMC article.
References
-
- Alfieri M. L., Micillo R., Panzella L., Crescenzi O., Oscurato S. L., Maddalena P., et al. . (2018a). Structural basis of polydopamine film formation: probing 5,6-dihydroxyindole-based eumelanin type units and the porphyrin issue. ACS Appl. Mater. Interfaces 10, 7670–7680. 10.1021/acsami.7b09662 - DOI - PubMed
LinkOut - more resources
Full Text Sources

