Electronic Effects of Substituents on fac-M(bpy-R)(CO)3 (M = Mn, Re) Complexes for Homogeneous CO2 Electroreduction
- PMID: 31231639
- PMCID: PMC6561311
- DOI: 10.3389/fchem.2019.00417
Electronic Effects of Substituents on fac-M(bpy-R)(CO)3 (M = Mn, Re) Complexes for Homogeneous CO2 Electroreduction
Abstract
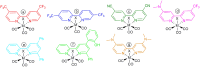
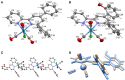
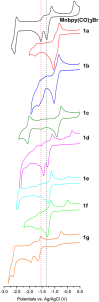
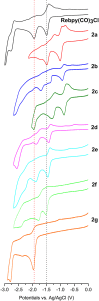
Synthesis and characterization of 14 new 2,2'-bipyridine metal complexes fac-M(bpy-R)(CO)3X (where M = Mn, X = Br or M = Re, X = Cl and R = -CF3, -CN, -Ph, -PhOH, -NMe2) are reported. The complexes have been characterized by NMR, IR spectroscopy and elemental analysis. Single crystal X-Ray diffraction structures have been solved for Re(dpbpy)(CO)3Cl (dpbpy = 4,6-diphenyl-2,2'-bipyridine) and Re(hpbpy)(CO)3Cl (hpbpy = 4-(2-hydroxy-phenyl)-6-phenyl-2,2'-bipyridine). Electrochemical behaviors of the complexes in acetonitrile under Ar and their catalytic performances for CO2 reduction with added water and MeOH have been investigated by cyclic voltammetry and controlled potential electrolysis. The role of the substituents on the electrochemical properties and the related over potentials required for CO2 transformation have been analyzed. The complexes carrying only electron withdrawing groups like -CF3, -CN totally lose their catalytic activities toward CO2 reduction, whereas the symmetric -NMe2 substituted and push-pull systems (containing both -NMe2 and -CF3) still display electrocatalytic current enhancement under CO2 atmosphere. The complexes carrying a phenyl or a phenol group in position 4 show catalytic behaviors similar to those of simple M-bpy systems. The only detected reduction product by GC analysis is CO: for example, fac-Re (bpy-4,4'-NMe2)(CO)3Cl gives CO with high faradic efficiency and a TON of 18 and 31, in absence of external proton source and with 5% MeOH, respectively. DFT calculations were carried out to highlight the electronic properties of the complexes; results are in agreement with experimental electrochemical data.
Keywords: CO2 electroreduction; DFT calculations; Mn complexes; Re complexes; bipy ligands; electron-donating; electron-withdrawing; homogeneous catalysis.
Figures


Similar articles
-
Local Proton Source in Electrocatalytic CO2 Reduction with [Mn(bpy-R)(CO)3 Br] Complexes.Chemistry. 2017 Apr 6;23(20):4782-4793. doi: 10.1002/chem.201605546. Epub 2017 Feb 22. Chemistry. 2017. PMID: 28106930
-
Graphene-Immobilized fac-Re(bipy)(CO)3Cl for Syngas Generation from Carbon Dioxide.ACS Appl Mater Interfaces. 2016 Feb 17;8(6):4192-8. doi: 10.1021/acsami.5b11958. Epub 2016 Feb 3. ACS Appl Mater Interfaces. 2016. PMID: 26799656
-
A Dicationic fac-Re(bpy)(CO)3Cl for CO2 Electroreduction at a Reduced Overpotential.Inorg Chem. 2023 May 22;62(20):7877-7889. doi: 10.1021/acs.inorgchem.3c00624. Epub 2023 May 9. Inorg Chem. 2023. PMID: 37159570
-
Molecular Catalysts with Intramolecular Re-O Bond for Electrochemical Reduction of Carbon Dioxide.Inorg Chem. 2020 Sep 8;59(17):12187-12199. doi: 10.1021/acs.inorgchem.0c01181. Epub 2020 Aug 17. Inorg Chem. 2020. PMID: 32804491 Free PMC article.
-
Comparisons of MN2S2vs. bipyridine as redox-active ligands to manganese and rhenium in (L-L)M'(CO)3Cl complexes.Dalton Trans. 2017 Apr 19;46(16):5175-5182. doi: 10.1039/c7dt00600d. Dalton Trans. 2017. PMID: 28322401
Cited by
-
Photocatalytic turnover of CO2 under visible light by [Re(CO)3(1-(1,10) phenanthroline-5-(4-nitro-naphthalimide))Cl] in tandem with the sacrificial donor BIH.RSC Adv. 2022 Feb 10;12(9):5080-5084. doi: 10.1039/d1ra08261b. eCollection 2022 Feb 10. RSC Adv. 2022. PMID: 35425589 Free PMC article.
-
Light-driven CO2 reduction with substituted imidazole-pyridine Re catalysts favoring formic acid production.RSC Adv. 2025 Apr 22;15(16):12547-12556. doi: 10.1039/d5ra01561h. eCollection 2025 Apr 16. RSC Adv. 2025. PMID: 40264887 Free PMC article.
-
Mechanistic Study of Tungsten Bipyridyl Tetracarbonyl Electrocatalysts for CO2 Fixation: Exploring the Roles of Explicit Proton Sources and Substituent Effects.Top Catal. 2022 Feb;65(1-4):325-340. doi: 10.1007/s11244-021-01529-7. Epub 2021 Nov 16. Top Catal. 2022. PMID: 37645456 Free PMC article.
-
Transition Metal Complexes as Catalysts for the Electroconversion of CO2 : An Organometallic Perspective.Angew Chem Int Ed Engl. 2021 May 17;60(21):11628-11686. doi: 10.1002/anie.202006988. Epub 2021 Jan 19. Angew Chem Int Ed Engl. 2021. PMID: 33464678 Free PMC article. Review.
-
How to rationally design homogeneous catalysts for efficient CO2 electroreduction?iScience. 2024 Jan 19;27(2):108973. doi: 10.1016/j.isci.2024.108973. eCollection 2024 Feb 16. iScience. 2024. PMID: 38327791 Free PMC article. Review.
References
-
- Agilent (2015). CrysAlis PRO. Yarnton: Agilent Technologies Ltd.
-
- Anderson P. A., Keene F. R., Horn E., Tiekink E. R. T. (1990). Ambidentate coordination of the tripyridyl ligands 2,2': 6',2”-terpyridyl, tris(2-pyridyl)-amine, tris(2-pyridyl)methane, and tris(2-pyridyl)phosphine to carbonylrhenium centres: structural and spectroscopic studies. Appl. Organomet. Chem. 4, 523–533. 10.1002/aoc.590040514 - DOI
-
- Becke A. D. (1993). Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652. 10.1063/1.464913 - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

