Non-canonical NLRP3 inflammasome activation and IL-1β signaling are necessary to L. amazonensis control mediated by P2X7 receptor and leukotriene B4
- PMID: 31233552
- PMCID: PMC6622556
- DOI: 10.1371/journal.ppat.1007887
Non-canonical NLRP3 inflammasome activation and IL-1β signaling are necessary to L. amazonensis control mediated by P2X7 receptor and leukotriene B4
Abstract
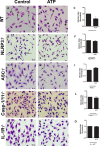
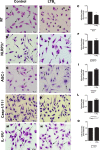
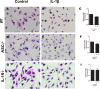
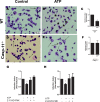
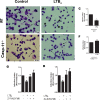
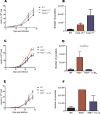
Leishmaniasis is a neglected tropical disease affecting millions of individuals worldwide. P2X7 receptor has been linked to the elimination of Leishmania amazonensis. Biological responses evoked by P2X7 receptor activation have been well-documented, including apoptosis, phagocytosis, cytokine release, such as IL-1β. It was demonstrated that NLRP3 inflammasome activation and IL-1β signaling participated in resistance against L. amazonensis. Furthermore, our group has shown that L. amazonensis elimination through P2X7 receptor activation depended on leukotriene B4 (LTB4) production and release. Therefore, we investigated whether L. amazonensis elimination by P2X7 receptor and LTB4 involved NLRP3 inflammasome activation and IL-1β signaling. We showed that macrophages from NLRP3-/-, ASC-/-, Casp-1/11-/-, gp91phox-/- , and IL-1R-/- mice treated with ATP or LTB4 did not decrease parasitic load as was observed in WT mice. When ASC-/- macrophages were treated with exogenous IL-1β, parasite killing was noted, however, we did not see parasitic load reduction in IL-1R-/- macrophages. Similarly, macrophages from P2X7 receptor-deficient mice treated with IL-1β also showed decreased parasitic load. In addition, when we infected Casp-11-/- macrophages, neither ATP nor LTB4 were able to reduce parasitic load, and Casp-11-/- mice were more susceptible to L. amazonensis infection than were WT mice. Furthermore, P2X7-/- L. amazonensis-infected mice locally treated with exogenous LTB4 showed resistance to infection, characterized by lower parasite load and smaller lesions compared to untreated P2X7-/- mice. A similar observation was noted when infected P2X7-/- mice were treated with IL-1β, i.e., lower parasite load and smaller lesions compared to P2X7-/- mice. These data suggested that L. amazonensis elimination mediated by P2X7 receptor and LTB4 was dependent on non-canonical NLRP3 inflammasome activation, ROS production, and IL-1β signaling.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
P2Y2 Receptor Induces L. amazonensis Infection Control in a Mechanism Dependent on Caspase-1 Activation and IL-1β Secretion.Mediators Inflamm. 2020 Oct 1;2020:2545682. doi: 10.1155/2020/2545682. eCollection 2020. Mediators Inflamm. 2020. PMID: 33061823 Free PMC article.
-
Leukotriene B4 modulates P2X7 receptor-mediated Leishmania amazonensis elimination in murine macrophages.J Immunol. 2014 May 15;192(10):4765-73. doi: 10.4049/jimmunol.1301058. Epub 2014 Apr 11. J Immunol. 2014. PMID: 24729618
-
Dectin-1 Activation during Leishmania amazonensis Phagocytosis Prompts Syk-Dependent Reactive Oxygen Species Production To Trigger Inflammasome Assembly and Restriction of Parasite Replication.J Immunol. 2017 Sep 15;199(6):2055-2068. doi: 10.4049/jimmunol.1700258. Epub 2017 Aug 7. J Immunol. 2017. PMID: 28784846
-
Silica-induced inflammasome activation in macrophages: role of ATP and P2X7 receptor.Immunobiology. 2015 Sep;220(9):1101-6. doi: 10.1016/j.imbio.2015.05.004. Epub 2015 May 18. Immunobiology. 2015. PMID: 26024943 Review.
-
Purinergic signaling: A new front-line determinant of resistance and susceptibility in leishmaniasis.Biomed J. 2022 Feb;45(1):109-117. doi: 10.1016/j.bj.2021.06.003. Epub 2021 Jun 24. Biomed J. 2022. PMID: 34175493 Free PMC article. Review.
Cited by
-
Cysteinyl-leukotrienes promote cutaneous Leishmaniasis control.Front Cell Infect Microbiol. 2023 Jun 12;13:1192800. doi: 10.3389/fcimb.2023.1192800. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37377641 Free PMC article.
-
Activation Pathways of Murine Macrophages by Lipophosphoglycan from Strains of Leishmania major (FV1 and LV39).ACS Infect Dis. 2024 Oct 11;10(10):3544-3552. doi: 10.1021/acsinfecdis.4c00295. Epub 2024 Sep 23. ACS Infect Dis. 2024. PMID: 39313410 Free PMC article.
-
P2Y2 Receptor Induces L. amazonensis Infection Control in a Mechanism Dependent on Caspase-1 Activation and IL-1β Secretion.Mediators Inflamm. 2020 Oct 1;2020:2545682. doi: 10.1155/2020/2545682. eCollection 2020. Mediators Inflamm. 2020. PMID: 33061823 Free PMC article.
-
Reconciling protective and pathogenic roles of the NLRP3 inflammasome in leishmaniasis.Immunol Rev. 2020 Sep;297(1):53-66. doi: 10.1111/imr.12886. Epub 2020 Jun 20. Immunol Rev. 2020. PMID: 32564424 Free PMC article. Review.
-
Critical Role for the NLRP3 Inflammasome in Mediating IL-1β Production in Shigella sonnei-Infected Macrophages.Front Immunol. 2020 Jun 3;11:1115. doi: 10.3389/fimmu.2020.01115. eCollection 2020. Front Immunol. 2020. PMID: 32582195 Free PMC article.
References
-
- Chaves SP, Torres-Santos EC, Marques C, Figliuolo VR, Persechini PM, Coutinho-Silva R, Rossi-Bergmann B (2009) Modulation of P2X(7) purinergic receptor in macrophages by Leishmania amazonensis and its role in parasite elimination. Microbes Infect 11: 842–849. S1286-4579(09)00103-8 [pii]; 10.1016/j.micinf.2009.05.001 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

