Inhibition of the Self-Assembly of Aβ and of Tau by Polyphenols: Mechanistic Studies
- PMID: 31234523
- PMCID: PMC6630797
- DOI: 10.3390/molecules24122316
Inhibition of the Self-Assembly of Aβ and of Tau by Polyphenols: Mechanistic Studies
Abstract
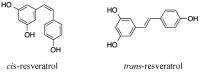
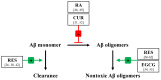
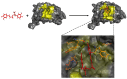
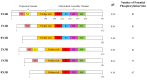
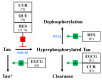
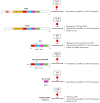
The amyloid-β (Aβ) peptide and tau protein are thought to play key neuropathogenic roles in Alzheimer's disease (AD). Both Aβ and tau self-assemble to form the two major pathological hallmarks of AD: amyloid plaques and neurofibrillary tangles, respectively. In this review, we show that naturally occurring polyphenols abundant in fruits, vegetables, red wine, and tea possess the ability to target pathways associated with the formation of assemblies of Aβ and tau. Polyphenols modulate the enzymatic processing of the amyloid-β precursor protein and inhibit toxic Aβ oligomerization by enhancing the clearance of Aβ42 monomer, modulating monomer-monomer interactions and remodeling oligomers to non-toxic forms. Additionally, polyphenols modulate tau hyperphosphorylation and inhibit tau β-sheet formation. The anti-Aβ-self-assembly and anti-tau-self-assembly effects of polyphenols increase their potential as preventive or therapeutic agents against AD, a complex disease that involves many pathological mechanisms.
Keywords: Alzheimer′s disease; amyloid assemblies; amyloid-β self-assembly; neurofibrillary tangles; polyphenols; tau hyperphosphorylation; tau self-assembly.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Lazo N.D., Maji S.K., Fradinger E.A., Bitan G., Teplow D.B. The amyloid-β protein. In: Sipe J.D., editor. Amyloid Proteins-the Beta Sheet Conformation and Disease. Wiley-VCH; Weinheim, Germany: 2005. pp. 385–491.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

