ALLENE OXIDE SYNTHASE and HYDROPEROXIDE LYASE, Two Non-Canonical Cytochrome P450s in Arabidopsis thaliana and Their Different Roles in Plant Defense
- PMID: 31234561
- PMCID: PMC6627107
- DOI: 10.3390/ijms20123064
ALLENE OXIDE SYNTHASE and HYDROPEROXIDE LYASE, Two Non-Canonical Cytochrome P450s in Arabidopsis thaliana and Their Different Roles in Plant Defense
Abstract
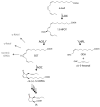
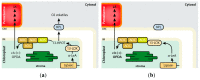
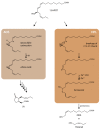
The channeling of metabolites is an essential step of metabolic regulation in all living organisms. Multifunctional enzymes with defined domains for metabolite compartmentalization are rare, but in many cases, larger assemblies forming multimeric protein complexes operate in defined metabolic shunts. In Arabidopsis thaliana, a multimeric complex was discovered that contains a 13-lipoxygenase and allene oxide synthase (AOS) as well as allene oxide cyclase. All three plant enzymes are localized in chloroplasts, contributing to the biosynthesis of jasmonic acid (JA). JA and its derivatives act as ubiquitous plant defense regulators in responses to both biotic and abiotic stresses. AOS belongs to the superfamily of cytochrome P450 enzymes and is named CYP74A. Another CYP450 in chloroplasts, hydroperoxide lyase (HPL, CYP74B), competes with AOS for the common substrate. The products of the HPL reaction are green leaf volatiles that are involved in the deterrence of insect pests. Both enzymes represent non-canonical CYP450 family members, as they do not depend on O2 and NADPH-dependent CYP450 reductase activities. AOS and HPL activities are crucial for plants to respond to different biotic foes. In this mini-review, we aim to summarize how plants make use of the LOX2-AOS-AOC2 complex in chloroplasts to boost JA biosynthesis over volatile production and how this situation may change in plant communities during mass ingestion by insect pests.
Keywords: allene oxide cyclase; allene oxide synthase; chloroplast envelope protein complex; hydroperoxide lyase; lipoxygenase; metabolite channeling; plant defense.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Substrate channeling in oxylipin biosynthesis through a protein complex in the plastid envelope of Arabidopsis thaliana.J Exp Bot. 2019 Mar 11;70(5):1483-1495. doi: 10.1093/jxb/erz015. J Exp Bot. 2019. PMID: 30690555 Free PMC article.
-
Tomato allene oxide synthase and fatty acid hydroperoxide lyase, two cytochrome P450s involved in oxylipin metabolism, are targeted to different membranes of chloroplast envelope.Plant Physiol. 2001 Jan;125(1):306-17. doi: 10.1104/pp.125.1.306. Plant Physiol. 2001. PMID: 11154338 Free PMC article.
-
Insect herbivores selectively suppress the HPL branch of the oxylipin pathway in host plants.Plant J. 2013 Feb;73(4):653-62. doi: 10.1111/tpj.12064. Epub 2012 Dec 28. Plant J. 2013. PMID: 23134585
-
Fatty acid hydroperoxide lyase: a plant cytochrome p450 enzyme involved in wound healing and pest resistance.Chembiochem. 2001 Aug 3;2(7-8):494-504. doi: 10.1002/1439-7633(20010803)2:7/8<494::AID-CBIC494>3.0.CO;2-1. Chembiochem. 2001. PMID: 11828481 Review.
-
Molecular mechanism of enzymatic allene oxide cyclization in plants.Plant Physiol Biochem. 2008 Mar;46(3):302-8. doi: 10.1016/j.plaphy.2007.12.007. Epub 2007 Dec 31. Plant Physiol Biochem. 2008. PMID: 18272375 Review.
Cited by
-
Cytochrome P450 Gene Families: Role in Plant Secondary Metabolites Production and Plant Defense.J Xenobiot. 2023 Jul 25;13(3):402-423. doi: 10.3390/jox13030026. J Xenobiot. 2023. PMID: 37606423 Free PMC article. Review.
-
Organizing the Chaos: Novel Insights into the Regulation of Z-3-Hexenal Production in Damaged Maize Leaves.Plants (Basel). 2024 Oct 3;13(19):2772. doi: 10.3390/plants13192772. Plants (Basel). 2024. PMID: 39409641 Free PMC article.
-
Abscisic acid receptors are involves in the Jasmonate signaling in Arabidopsis.Plant Signal Behav. 2021 Oct 3;16(10):1948243. doi: 10.1080/15592324.2021.1948243. Epub 2021 Jul 5. Plant Signal Behav. 2021. PMID: 34224307 Free PMC article.
-
Genome and transcriptome analysis to understand the role diversification of cytochrome P450 gene under excess nitrogen treatment.BMC Plant Biol. 2021 Oct 6;21(1):447. doi: 10.1186/s12870-021-03224-x. BMC Plant Biol. 2021. PMID: 34615481 Free PMC article.
-
Structure and Function of the Cytochrome P450 Monooxygenase Cinnamate 4-hydroxylase from Sorghum bicolor.Plant Physiol. 2020 Jul;183(3):957-973. doi: 10.1104/pp.20.00406. Epub 2020 Apr 24. Plant Physiol. 2020. PMID: 32332088 Free PMC article.
References
-
- Corey E.J., D’Alarcao M., Matsuda S.P.T., Lansbury P.T., Jr., Yamada Y. Intermediacy of 8-(R)-HPETE in the conversion of arachidonic acid to pre-clavulone a by Clavularia viridis. Implications for the biosynthesis of marine prostanoids. J. Am. Chem. Soc. 1987;109:289–290. doi: 10.1021/ja00235a053. - DOI
-
- Corey E.J., Matsuda S.P.T., Nagata R., Cleaver M.B. Biosynthesis of 8-R-HPETE and preclavulone-A from arachidonate in several species of caribbean coral. A widespread route to marine prostanoids. Tetrahedron Lett. 1988;29:2555–2558. doi: 10.1016/S0040-4039(00)86110-9. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

