Chemical depletion of phagocytic immune cells in Anopheles gambiae reveals dual roles of mosquito hemocytes in anti- Plasmodium immunity
- PMID: 31235594
- PMCID: PMC6628807
- DOI: 10.1073/pnas.1900147116
Chemical depletion of phagocytic immune cells in Anopheles gambiae reveals dual roles of mosquito hemocytes in anti- Plasmodium immunity
Abstract
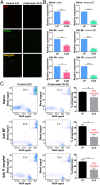
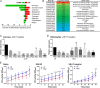
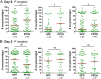
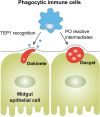
Mosquito immunity is composed of both cellular and humoral factors that provide protection from invading pathogens. Immune cells known as hemocytes, have been intricately associated with phagocytosis and innate immune signaling. However, the lack of genetic tools has limited hemocyte study despite their importance in mosquito anti-Plasmodium immunity. To address these limitations, we employ the use of a chemical-based treatment to deplete phagocytic immune cells in Anopheles gambiae, demonstrating the role of phagocytes in complement recognition and prophenoloxidase production that limit the ookinete and oocyst stages of malaria parasite development, respectively. Through these experiments, we also define specific subtypes of phagocytic immune cells in An. gambiae, providing insights beyond the morphological characteristics that traditionally define mosquito hemocyte populations. Together, this study represents a significant advancement in our understanding of the roles of mosquito phagocytes in mosquito vector competence and demonstrates the utility of clodronate liposomes as an important tool in the study of invertebrate immunity.
Keywords: Anopheles gambiae; hemocytes; innate immunity; malaria; phagocyte depletion.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Anopheles gambiae phagocytic hemocytes promote Plasmodium falciparum infection by regulating midgut epithelial integrity.Nat Commun. 2025 Feb 8;16(1):1465. doi: 10.1038/s41467-025-56313-y. Nat Commun. 2025. PMID: 39920122 Free PMC article.
-
Late-phase immune responses limiting oocyst survival are independent of TEP1 function yet display strain specific differences in Anopheles gambiae.Parasit Vectors. 2017 Aug 1;10(1):369. doi: 10.1186/s13071-017-2308-0. Parasit Vectors. 2017. PMID: 28764765 Free PMC article.
-
Single-cell analysis of mosquito hemocytes identifies signatures of immune cell subtypes and cell differentiation.Elife. 2021 Jul 28;10:e66192. doi: 10.7554/eLife.66192. Elife. 2021. PMID: 34318744 Free PMC article.
-
NF-κB-Like Signaling Pathway REL2 in Immune Defenses of the Malaria Vector Anopheles gambiae.Front Cell Infect Microbiol. 2017 Jun 21;7:258. doi: 10.3389/fcimb.2017.00258. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28680852 Free PMC article. Review.
-
How does Anopheles gambiae kill malaria parasites?Parassitologia. 1999 Sep;41(1-3):169-75. Parassitologia. 1999. PMID: 10697851 Review.
Cited by
-
Prostaglandin E2 Signaling Mediates Oenocytoid Immune Cell Function and Lysis, Limiting Bacteria and Plasmodium Oocyst Survival in Anopheles gambiae.Front Immunol. 2021 Aug 16;12:680020. doi: 10.3389/fimmu.2021.680020. eCollection 2021. Front Immunol. 2021. PMID: 34484178 Free PMC article.
-
The 20-hydroxyecdysone agonist, halofenozide, promotes anti-Plasmodium immunity in Anopheles gambiae via the ecdysone receptor.Sci Rep. 2020 Dec 3;10(1):21084. doi: 10.1038/s41598-020-78280-8. Sci Rep. 2020. PMID: 33273588 Free PMC article.
-
Anopheles gambiae phagocytic hemocytes promote Plasmodium falciparum infection by regulating midgut epithelial integrity.Nat Commun. 2025 Feb 8;16(1):1465. doi: 10.1038/s41467-025-56313-y. Nat Commun. 2025. PMID: 39920122 Free PMC article.
-
The CLIP-domain serine protease CLIPC9 regulates melanization downstream of SPCLIP1, CLIPA8, and CLIPA28 in the malaria vector Anopheles gambiae.PLoS Pathog. 2020 Oct 12;16(10):e1008985. doi: 10.1371/journal.ppat.1008985. eCollection 2020 Oct. PLoS Pathog. 2020. PMID: 33045027 Free PMC article.
-
A novel Galleria mellonella experimental model for zoonotic pathogen Brucella.Virulence. 2023 Dec;14(1):2268496. doi: 10.1080/21505594.2023.2268496. Epub 2023 Oct 23. Virulence. 2023. PMID: 37817444 Free PMC article.
References
-
- Salzet M., Vertebrate innate immunity resembles a mosaic of invertebrate immune responses. Trends Immunol. 22, 285–288 (2001). - PubMed
-
- Williams M. J., Drosophila hemopoiesis and cellular immunity. J. Immunol. 178, 4711–4716 (2007). - PubMed
-
- Honti V., Csordás G., Kurucz É., Márkus R., Andó I., The cell-mediated immunity of Drosophila melanogaster: Hemocyte lineages, immune compartments, microanatomy and regulation. Dev. Comp. Immunol. 42, 47–56 (2014). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

