Mathematical modeling of sleep state dynamics in a rodent model of shift work
- PMID: 31236510
- PMCID: PMC6584688
- DOI: 10.1016/j.nbscr.2018.04.002
Mathematical modeling of sleep state dynamics in a rodent model of shift work
Abstract
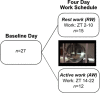
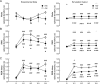
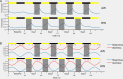
Millions of people worldwide are required to work when their physiology is tuned for sleep. By forcing wakefulness out of the body's normal schedule, shift workers face numerous adverse health consequences, including gastrointestinal problems, sleep problems, and higher rates of some diseases, including cancers. Recent studies have developed protocols to simulate shift work in rodents with the intention of assessing the effects of night-shift work on subsequent sleep (Grønli et al., 2017). These studies have already provided important contributions to the understanding of the metabolic consequences of shift work (Arble et al., 2015; Marti et al., 2016; Opperhuizen et al., 2015) and sleep-wake-specific impacts of night-shift work (Grønli et al., 2017). However, our understanding of the causal mechanisms underlying night-shift-related sleep disturbances is limited. In order to advance toward a mechanistic understanding of sleep disruption in shift work, we model these data with two different approaches. First we apply a simple homeostatic model to quantify differences in the rates at which sleep need, as measured by slow wave activity during slow wave sleep (SWS) rises and falls. Second, we develop a simple and novel mathematical model of rodent sleep and use it to investigate the timing of sleep in a simulated shift work protocol (Grønli et al., 2017). This mathematical framework includes the circadian and homeostatic processes of the two-process model, but additionally incorporates a stochastic process to model the polyphasic nature of rodent sleep. By changing only the time at which the rodents are forced to be awake, the model reproduces some key experimental results from the previous study, including correct proportions of time spent in each stage of sleep as a function of circadian time and the differences in total wake time and SWS bout durations in the rodents representing night-shift workers and those representing day-shift workers. Importantly, the model allows for deeper insight into circadian and homeostatic influences on sleep timing, as it demonstrates that the differences in SWS bout duration between rodents in the two shifts is largely a circadian effect. Our study shows the importance of mathematical modeling in uncovering mechanisms behind shift work sleep disturbances and it begins to lay a foundation for future mathematical modeling of sleep in rodents.
Figures





Similar articles
-
A Rodent Model of Night-Shift Work Induces Short-Term and Enduring Sleep and Electroencephalographic Disturbances.J Biol Rhythms. 2017 Feb;32(1):48-63. doi: 10.1177/0748730416675460. Epub 2016 Dec 14. J Biol Rhythms. 2017. PMID: 28013579
-
Hormonal and pharmacological manipulation of the circadian clock: recent developments and future strategies.Sleep. 2000 May 1;23 Suppl 3:S77-85. Sleep. 2000. PMID: 10809190 Review.
-
A mathematical model of ultradian sleep-wake regulation in rodents.Annu Int Conf IEEE Eng Med Biol Soc. 2016 Aug;2016:1656-1659. doi: 10.1109/EMBC.2016.7591032. Annu Int Conf IEEE Eng Med Biol Soc. 2016. PMID: 28268648
-
Day and night shift schedules are associated with lower sleep quality in Evening-types.Chronobiol Int. 2015 Jun;32(5):627-36. doi: 10.3109/07420528.2015.1033425. Epub 2015 Jun 2. Chronobiol Int. 2015. PMID: 26035480
-
Physiologically-based modeling of sleep-wake regulatory networks.Math Biosci. 2014 Apr;250:54-68. doi: 10.1016/j.mbs.2014.01.012. Epub 2014 Feb 11. Math Biosci. 2014. PMID: 24530893 Review.
Cited by
-
Chronic dietary supplementation with nicotinamide riboside reduces sleep need in the laboratory mouse.Sleep Adv. 2023 Dec 20;4(1):zpad044. doi: 10.1093/sleepadvances/zpad044. eCollection 2023. Sleep Adv. 2023. PMID: 38152423 Free PMC article.
-
Recent advances in modeling sleep: from the clinic to society and disease.Curr Opin Physiol. 2020 Jun;15:37-46. doi: 10.1016/j.cophys.2019.12.001. Epub 2019 Dec 13. Curr Opin Physiol. 2020. PMID: 34485783 Free PMC article.
-
Cognitive function and brain plasticity in a rat model of shift work: role of daily rhythms, sleep and glucocorticoids.Sci Rep. 2020 Aug 4;10(1):13141. doi: 10.1038/s41598-020-69969-x. Sci Rep. 2020. PMID: 32753733 Free PMC article.
References
-
- Achermann P., Borbély A.A. Simulation of daytime vigilance by the additive interaction of a homeostatic and a circadian process. Biol. Cybern. 1994;71(2):115–121. - PubMed
-
- Akerstedt T., Torsvall L., Gillberg M. Sleepiness in shiftwork. A review with emphasis on continuous monitoring of EEG and EOG. Chrono-. Int. 1987;4(2):129–140. - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

