Broad Lipidomic and Transcriptional Changes of Prophylactic PEA Administration in Adult Mice
- PMID: 31244590
- PMCID: PMC6580993
- DOI: 10.3389/fnins.2019.00527
Broad Lipidomic and Transcriptional Changes of Prophylactic PEA Administration in Adult Mice
Abstract
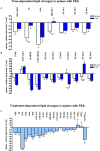
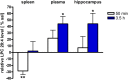
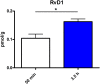
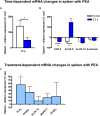
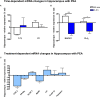
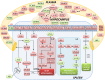
Beside diverse therapeutic properties of palmitoylethanolamide (PEA) including: neuroprotection, inflammation and pain alleviation, prophylactic effects have also been reported in animal models of infections, inflammation, and neurological diseases. The availability of PEA as (ultra)micronized nutraceutical formulations with reportedly no side effects, renders it accordingly an appealing candidate in human preventive care, such as in population at high risk of disease development or for healthy aging. PEA's mode of action is multi-facetted. Consensus exists that PEA's effects are primarily modulated by the peroxisome proliferator-activated receptor alpha (PPARα) and that PEA-activated PPARα has a pleiotropic effect on lipid metabolism, inflammation gene networks, and host defense mechanisms. Yet, an exhaustive view of how the prophylactic PEA administration changes the lipid signaling in brain and periphery, thereby eliciting a beneficial response to various negative stimuli remains still elusive. We therefore, undertook a broad lipidomic and transcriptomic study in brain and spleen of adult mice to unravel the positive molecular phenotype rendered by prophylactic PEA. We applied a tissue lipidomic and transcriptomic approach based on simultaneous extraction and subsequent targeted liquid chromatography-multiple reaction monitoring (LC-MRM) and mRNA analysis by qPCR, respectively. We targeted lipids of COX-, LOX- and CYP450 pathways, respectively, membrane phospholipids, lipid products of cPLA2, and free fatty acids, along with various genes involved in their biosynthesis and function. Additionally, plasma lipidomics was applied to reveal circulatory consequences and/or reflection of PEA's action. We found broad, distinct, and several previously unknown tissue transcriptional regulations of inflammatory pathways. In hippocampus also a PEA-induced transcriptional regulation of neuronal activity and excitability was evidenced. A massive downregulation of membrane lipid levels in the splenic tissue of the immune system with a consequent shift towards pro-resolving lipid environment was also detected. Plasma lipid pattern reflected to a large extent the hippocampal and splenic lipidome changes, highlighting the value of plasma lipidomics to monitor effects of nutraceutical PEA administration. Altogether, these findings contribute new insights into PEA's molecular mechanism and helps answering the questions, how PEA prepares the body for insults and what are the "good lipids" that underlie this action.
Keywords: PEA; PUFAs; endocannabinoids; inflammation; mRNA; phospholipids; targeted lipidomics.
Figures



References
-
- Artukoglu B. B., Beyer C., Zuloff-Shani A., Brener E., Bloch M. (2017). Efficacy of palmitoylethanolamide for pain: a meta-analysis. Pain Physician 20 353–362. - PubMed
LinkOut - more resources
Full Text Sources

