The Role of Wnt and R-spondin in the Stomach During Health and Disease
- PMID: 31248166
- PMCID: PMC6631168
- DOI: 10.3390/biomedicines7020044
The Role of Wnt and R-spondin in the Stomach During Health and Disease
Abstract
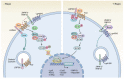
The Wnt signaling pathway is one of the most prominent developmental signals. In addition to its functions in development, there is emerging evidence that it is also crucial for various organ functions in adult organisms, where Wnt signaling controls tissue stem cell behavior, proliferation and differentiation. Deregulation of Wnt signaling is involved in various pathological conditions and has been linked to malignant tissue transformation in different organ systems. The study of the Wnt signaling pathway has revealed a complex regulatory network that tightly balances the quality and strength of Wnt signaling in tissues. In this context, R-spondins are secreted proteins that stabilize Wnt receptors and enhance Wnt signaling. In this review we focus on new insights into the regulatory function of Wnt and R-spondin signaling in the stomach. In addition to its function in the healthy state, we highlight the connection between Wnt signaling and infection with Helicobacter pylori (H. pylori), a pathogen that colonizes the stomach and is the main risk factor for gastric cancer. In addition to experimental data that link Wnt signaling to carcinogenesis, we discuss that Wnt signaling is affected in a substantial proportion of patients with gastric cancer, and provide examples for potential clinical implications for altered Wnt signaling in gastric cancer.
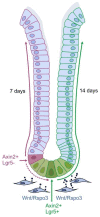
Keywords: Axin2; Helicobacter pylori; Lgr5+ stem cells; R-spondin; Wnt signaling; gastric cancer; stem cells.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

