Monocytes, Macrophages, and Metabolic Disease in Atherosclerosis
- PMID: 31249530
- PMCID: PMC6584106
- DOI: 10.3389/fphar.2019.00666
Monocytes, Macrophages, and Metabolic Disease in Atherosclerosis
Abstract
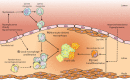
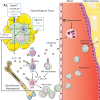
Atherosclerotic cardiovascular disease (CVD) is a lipid-driven chronic inflammatory disease, in which macrophages are responsible for taking up these lipids and driving disease progression. Over the years, we and others have uncovered key pathways that regulate macrophage number/function and identified how metabolic disorders such as diabetes and obesity, which are common risk factors for CVD, exacerbate these pathways. This ultimately accelerates the progression of atherosclerosis and hinders atherosclerotic regression. In this review, we discuss the different types of macrophages, from monocyte-derived macrophages, local macrophage proliferation, to macrophage-like vascular smooth muscle cells, that contribute to atherosclerosis as well as myeloid-derived suppressor cells that may have anti-atherogenic effects. We will also discuss how diabetes and obesity influence plaque macrophage accumulation and monocyte production (myelopoiesis) to promote atherogenesis as well as an exciting therapeutic target, S100A8/A9, which mediates myelopoiesis in response to both diabetes and obesity, shown to be effective in reducing atherosclerosis in pre-clinical models of diabetes.
Keywords: atherosclerosis; diabetes; macrophage; monocyte; obesity.
Figures
References
-
- Barr E. L., Zimmet P. Z., Welborn T. A., Jolley D., Magliano D. J., Dunstan D. W., et al. (2007). Risk of cardiovascular and all-cause mortality in individuals with diabetes mellitus, impaired fasting glucose, and impaired glucose tolerance: the Australian Diabetes, Obesity, and Lifestyle Study (AusDiab). Circulation 116 (2), 151–157. 10.1161/CIRCULATIONAHA.106.685628 - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

