How, When, and Where Do Human β-Cells Regenerate?
- PMID: 31250214
- PMCID: PMC6986204
- DOI: 10.1007/s11892-019-1176-8
How, When, and Where Do Human β-Cells Regenerate?
Abstract
Purpose of review: Pancreatic β-cells play a critical role in whole-body glucose homeostasis by regulating the release of insulin in response to minute by minute alterations in metabolic demand. As such, β-cells are staunchly resilient but there are circumstances where they can become functionally compromised or physically lost due to pathophysiological changes which culminate in overt hyperglycemia and diabetes.
Recent findings: In humans, β-cell mass appears to be largely defined in the postnatal period and this early replicative and generative phase is followed by a refractory state which persists throughout life. Despite this, efforts to identify physiological and pharmacological factors which might re-initiate β-cell replication (or cause the replenishment of β-cells by neogenesis or transdifferentiation) are beginning to bear fruit. Controlled manipulation of β-cell mass in humans still represents a holy grail for therapeutic intervention in diabetes, but progress is being made which may lead to ultimate success.
Keywords: Diabetes; Islets of Langerhans; Ki67; Proliferation; Transdifferentiation; β-Cell mass.
Conflict of interest statement
Figures
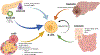

References
-
- Shields BM, Freathy RM, Hattersley AT. Genetic influences on the association between fetal growth and susceptibility to type 2 diabetes. J Dev Orig Health Dis. 2010;1(2):96–105. - PubMed
-
- Spyer G, Macleod KM, Shepherd M, Ellard S, Hattersley AT. Pregnancy outcome in patients with raised blood glucose due to a heterozygous glucokinase gene mutation. Diabet Med. 2009;26(1):14–8. - PubMed
-
- Jennings RE, Berry AA, Strutt JP, Gerrard DT, Hanley NA. Human pancreas development. Development. 2015;142(18): 3126–37. - PubMed
-
- Cnop M, Igoillo-Esteve M, Hughes SJ, Walker JN, Cnop I, Clark A. Longevity of human islet alpha and beta cells. Diabetes Obes Metab. 2011;13(Suppl 1):39–46. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

