MFS transporter from Botrytis cinerea provides tolerance to glucosinolate-breakdown products and is required for pathogenicity
- PMID: 31253809
- PMCID: PMC6599007
- DOI: 10.1038/s41467-019-10860-3
MFS transporter from Botrytis cinerea provides tolerance to glucosinolate-breakdown products and is required for pathogenicity
Abstract
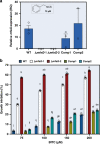
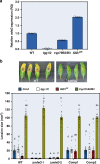
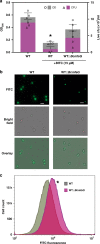
Glucosinolates accumulate mainly in cruciferous plants and their hydrolysis-derived products play important roles in plant resistance against pathogens. The pathogen Botrytis cinerea has variable sensitivity to glucosinolates, but the mechanisms by which it responds to them are mostly unknown. Exposure of B. cinerea to glucosinolate-breakdown products induces expression of the Major Facilitator Superfamily transporter, mfsG, which functions in fungitoxic compound efflux. Inoculation of B. cinerea on wild-type Arabidopsis thaliana plants induces mfsG expression to higher levels than on glucosinolate-deficient A. thaliana mutants. A B. cinerea strain lacking functional mfsG transporter is deficient in efflux ability. It accumulates more isothiocyanates (ITCs) and is therefore more sensitive to this compound in vitro; it is also less virulent to glucosinolates-containing plants. Moreover, mfsG mediates ITC efflux in Saccharomyces cerevisiae cells, thereby conferring tolerance to ITCs in the yeast. These findings suggest that mfsG transporter is a virulence factor that increases tolerance to glucosinolates.
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
The ABC transporter BcatrB from Botrytis cinerea exports camalexin and is a virulence factor on Arabidopsis thaliana.Plant J. 2009 May;58(3):499-510. doi: 10.1111/j.1365-313X.2009.03794.x. Epub 2008 Jan 18. Plant J. 2009. PMID: 19154205
-
The effects of glucosinolates and their breakdown products on necrotrophic fungi.PLoS One. 2013 Aug 5;8(8):e70771. doi: 10.1371/journal.pone.0070771. Print 2013. PLoS One. 2013. PMID: 23940639 Free PMC article.
-
Comparing Fungal Sensitivity to Isothiocyanate Products on Different Botrytis spp.Plants (Basel). 2024 Mar 7;13(6):756. doi: 10.3390/plants13060756. Plants (Basel). 2024. PMID: 38592765 Free PMC article.
-
Botrytis small RNA Bc-siR37 suppresses plant defense genes by cross-kingdom RNAi.RNA Biol. 2017 Apr 3;14(4):421-428. doi: 10.1080/15476286.2017.1291112. Epub 2017 Mar 7. RNA Biol. 2017. PMID: 28267415 Free PMC article. Review.
-
Effects of Glucosinolate-Derived Isothiocyanates on Fungi: A Comprehensive Review on Direct Effects, Mechanisms, Structure-Activity Relationship Data and Possible Agricultural Applications.J Fungi (Basel). 2021 Jul 6;7(7):539. doi: 10.3390/jof7070539. J Fungi (Basel). 2021. PMID: 34356918 Free PMC article. Review.
Cited by
-
Virulence-related metabolism is activated in Botrytis cinerea mostly in the interaction with tolerant green grapes that remain largely unaffected in contrast with susceptible green grapes.Hortic Res. 2022 Sep 21;9:uhac217. doi: 10.1093/hr/uhac217. eCollection 2022. Hortic Res. 2022. PMID: 36479580 Free PMC article.
-
Transcriptional differentiation driving Cucumis sativus-Botrytis cinerea interactions based on the Skellam model and Bayesian networks.AMB Express. 2021 Oct 20;11(1):138. doi: 10.1186/s13568-021-01296-4. AMB Express. 2021. PMID: 34669064 Free PMC article.
-
Understanding Efflux-Mediated Multidrug Resistance in Botrytis cinerea for Improved Management of Fungicide Resistance.Microb Biotechnol. 2025 Mar;18(3):e70074. doi: 10.1111/1751-7915.70074. Microb Biotechnol. 2025. PMID: 40133238 Free PMC article. Review.
-
BcWRKY33A Enhances Resistance to Botrytis cinerea via Activating BcMYB51-3 in Non-Heading Chinese Cabbage.Int J Mol Sci. 2022 Jul 26;23(15):8222. doi: 10.3390/ijms23158222. Int J Mol Sci. 2022. PMID: 35897830 Free PMC article.
-
Molecular Characterisation of the Peroxidase Gene Family in Botrytis cinerea and the Role of BcPRD7 in Virulence.Mol Plant Pathol. 2025 Apr;26(4):e70079. doi: 10.1111/mpp.70079. Mol Plant Pathol. 2025. PMID: 40165570 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

