The Ratio of Factor VIIa:Tissue Factor Content within Microvesicles Determines the Differential Influence on Endothelial Cells
- PMID: 31259295
- PMCID: PMC6598090
- DOI: 10.1055/s-0039-1688934
The Ratio of Factor VIIa:Tissue Factor Content within Microvesicles Determines the Differential Influence on Endothelial Cells
Abstract
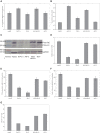
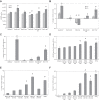
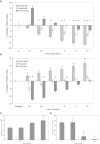
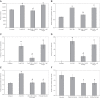
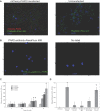
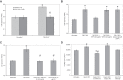
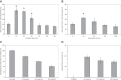
Tissue factor (TF)-positive microvesicles from various sources can promote cellular proliferation or alternatively induce apoptosis, but the determining factors are unknown. In this study the hypothesis that the ratio of fVIIa:TF within microvesicles determines this outcome was examined. Microvesicles were isolated from HepG2, BxPC-3, 786-O, MDA-MB-231, and MCF-7 cell lines and microvesicle-associated fVIIa and TF antigen and activity levels were measured. Human coronary artery endothelial cells (HCAECs) were incubated with these purified microvesicles, or with combinations of fVIIa-recombinant TF, and cell proliferation/apoptosis was measured. Additionally, by expressing mCherry-PAR2 on HCAEC surface, PAR2 activation was quantified. Finally, the activation of PAR2 on HCAEC or the activities of TF and fVIIa in microvesicles were blocked prior to addition of microvesicles to cells. The purified microvesicles exhibited a range of fVIIa:TF ratios with HepG2 and 786-O cells having the highest (54:1) and lowest (10:1) ratios, respectively. The reversal from proapoptotic to proliferative was estimated to occur at a fVIIa:TF molar ratio of 15:1, but HCAEC could not be rescued at higher TF concentrations. The purified microvesicles induced HCAEC proliferation or apoptosis according to this ruling. Blocking PAR2 activation on HCAEC, or inhibiting fVIIa or TF-procoagulant function on microvesicles prevented the influence on HCAEC. Finally, incubation of HCAEC with recombinant TF resulted in increased surface exposure of fVII. The induction of cell proliferation or apoptosis by TF-positive microvesicles is dependent on the ratio of fVIIa:TF and involves the activation of PAR2. At lower TF concentrations, fVIIa can counteract the proapoptotic stimulus and induce proliferation.
Keywords: apoptosis; cell proliferation; factor VIIa; microvesicles; protease-activated receptor-2; tissue factor.
Conflict of interest statement
Figures
Similar articles
-
Factor VIIa Regulates the Level of Cell-Surface Tissue Factor through Separate but Cooperative Mechanisms.Cancers (Basel). 2021 Jul 23;13(15):3718. doi: 10.3390/cancers13153718. Cancers (Basel). 2021. PMID: 34359618 Free PMC article.
-
Endothelial-derived microvesicles promote pro-migratory cross-talk with smooth muscle cells by a mechanism requiring tissue factor and PAR2 activation.Front Cardiovasc Med. 2024 Jun 20;11:1365008. doi: 10.3389/fcvm.2024.1365008. eCollection 2024. Front Cardiovasc Med. 2024. PMID: 38966751 Free PMC article.
-
Apixaban Suppresses the Release of TF-Positive Microvesicles and Restrains Cancer Cell Proliferation through Directly Inhibiting TF-fVIIa Activity.Thromb Haemost. 2019 Sep;119(9):1419-1432. doi: 10.1055/s-0039-1692682. Epub 2019 Jul 2. Thromb Haemost. 2019. PMID: 31266079
-
Microarray studies of factor VIIa-activated cancer cells.Thromb Res. 2008;122 Suppl 1:S11-3. doi: 10.1016/S0049-3848(08)70011-2. Thromb Res. 2008. PMID: 18691491 Review.
-
The action of high-dose factor VIIa (FVIIa) in a cell-based model of hemostasis.Dis Mon. 2003 Jan;49(1):14-21. doi: 10.1053/mda.2003.29504b. Dis Mon. 2003. PMID: 12525825 Review.
Cited by
-
Porphyromonas gingivalis triggers the shedding of inflammatory endothelial microvesicles that act as autocrine effectors of endothelial dysfunction.Sci Rep. 2020 Feb 4;10(1):1778. doi: 10.1038/s41598-020-58374-z. Sci Rep. 2020. PMID: 32019950 Free PMC article.
-
Accumulation of tissue factor in endothelial cells promotes cellular apoptosis through over-activation of Src1 and involves β1-integrin signalling.Apoptosis. 2020 Feb;25(1-2):29-41. doi: 10.1007/s10495-019-01576-2. Apoptosis. 2020. PMID: 31654241 Free PMC article.
-
Factor VIIa Regulates the Level of Cell-Surface Tissue Factor through Separate but Cooperative Mechanisms.Cancers (Basel). 2021 Jul 23;13(15):3718. doi: 10.3390/cancers13153718. Cancers (Basel). 2021. PMID: 34359618 Free PMC article.
-
Activation of PAR2 by tissue factor induces the release of the PTEN from MAGI proteins and regulates PTEN and Akt activities.Sci Rep. 2020 Dec 1;10(1):20908. doi: 10.1038/s41598-020-77963-6. Sci Rep. 2020. PMID: 33262514 Free PMC article.
-
Tissue factor signalling modifies the expression and regulation of G1/S checkpoint regulators: Implications during injury and prolonged inflammation.Mol Med Rep. 2025 Feb;31(2):39. doi: 10.3892/mmr.2024.13404. Epub 2024 Nov 29. Mol Med Rep. 2025. PMID: 39611476 Free PMC article.
References
-
- Kirchhofer D, Nemerson Y. Initiation of blood coagulation: the tissue factor/factor VIIa complex. Curr Opin Biotechnol. 1996;7(04):386–391. - PubMed
-
- Edgington T S, Dickinson C D, Ruf W. The structural basis of function of the TF. VIIa complex in the cellular initiation of coagulation. Thromb Haemost. 1997;78(01):401–405. - PubMed
-
- Morel O, Toti F, Hugel B et al.Procoagulant microparticles: disrupting the vascular homeostasis equation? Arterioscler Thromb Vasc Biol. 2006;26(12):2594–2604. - PubMed
-
- Freyssinet J M, Toti F. Formation of procoagulant microparticles and properties. Thromb Res. 2010;125 01:S46–S48. - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

