mGlu5 Positive Allosteric Modulators Facilitate Long-Term Potentiation via Disinhibition Mediated by mGlu5-Endocannabinoid Signaling
- PMID: 31259318
- PMCID: PMC6591772
- DOI: 10.1021/acsptsci.9b00017
mGlu5 Positive Allosteric Modulators Facilitate Long-Term Potentiation via Disinhibition Mediated by mGlu5-Endocannabinoid Signaling
Abstract
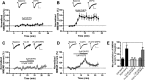
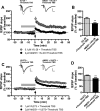
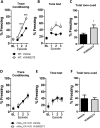
Metabotropic glutamate (mGlu) receptor type 5 (mGlu5) positive allosteric modulators (PAMs) enhance hippocampal long-term potentiation (LTP) and have cognition-enhancing effects in animal models. These effects were initially thought to be mediated by potentiation of mGlu5 modulation of N-methyl-d-aspartate receptor (NMDAR) currents. However, a biased mGlu5 PAM that potentiates Gαq-dependent mGlu5 signaling, but not mGlu5 modulation of NMDAR currents, retains cognition-enhancing effects in animal models, suggesting that potentiation of NMDAR currents is not required for these in vivo effects of mGlu5 PAMs. However, it is not clear whether the potentiation of NMDAR currents is critical for the ability of mGlu5 PAMs to enhance hippocampal LTP. We now report the characterization of effects of two structurally distinct mGlu5 PAMs, VU-29 and VU0092273, on NMDAR currents and hippocampal LTP. As with other mGlu5 PAMs that do not display observable bias for potentiation of NMDAR currents, VU0092273 enhanced both mGlu5 modulation of NMDAR currents and induction of LTP at the hippocampal Schaffer collateral (SC)-CA1 synapse. In contrast, VU-29 did not potentiate mGlu5 modulation of NMDAR currents but induced robust potentiation of hippocampal LTP. Interestingly, both VU-29 and VU0092273 suppressed evoked inhibitory postsynaptic currents (eIPSCs) in CA1 pyramidal cells, and this effect was blocked by the cannabinoid receptor type 1 (CB1) antagonist AM251. Furthermore, AM251 blocked the ability of both mGlu5 PAMs to enhance LTP. Finally, both PAMs failed to enhance LTP in mice with the restricted genetic deletion of mGlu5 in CA1 pyramidal cells. Taken together with previous findings, these results suggest that enhancement of LTP by mGlu5 PAMs does not depend on mGlu5 modulation of NMDAR currents but is mediated by a previously established mechanism in which mGlu5 in CA1 pyramidal cells induces endocannabinoid release and CB1-dependent disinhibition.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




Similar articles
-
Role of mGlu5 Receptors and Inhibitory Neurotransmission in M1 Dependent Muscarinic LTD in the Prefrontal Cortex: Implications in Schizophrenia.ACS Chem Neurosci. 2017 Oct 18;8(10):2254-2265. doi: 10.1021/acschemneuro.7b00167. Epub 2017 Aug 9. ACS Chem Neurosci. 2017. PMID: 28679049 Free PMC article.
-
Biased mGlu5-Positive Allosteric Modulators Provide In Vivo Efficacy without Potentiating mGlu5 Modulation of NMDAR Currents.Neuron. 2015 May 20;86(4):1029-1040. doi: 10.1016/j.neuron.2015.03.063. Epub 2015 Apr 30. Neuron. 2015. PMID: 25937172 Free PMC article.
-
Activation of Group II Metabotropic Glutamate Receptors Promotes LTP Induction at Schaffer Collateral-CA1 Pyramidal Cell Synapses by Priming NMDA Receptors.J Neurosci. 2016 Nov 9;36(45):11521-11531. doi: 10.1523/JNEUROSCI.1519-16.2016. J Neurosci. 2016. PMID: 27911756 Free PMC article.
-
Pure Positive Allosteric Modulators (PAMs) of mGlu5 with Competitive MPEP-Site Interaction.2011 Oct 31 [updated 2013 Mar 7]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. 2011 Oct 31 [updated 2013 Mar 7]. In: Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2010–. PMID: 23762950 Free Books & Documents. Review.
-
Long-term potentiation and the role of N-methyl-D-aspartate receptors.Brain Res. 2015 Sep 24;1621:5-16. doi: 10.1016/j.brainres.2015.01.016. Epub 2015 Jan 22. Brain Res. 2015. PMID: 25619552 Free PMC article. Review.
Cited by
-
The Neuroprotective Effects of mGlu1 Receptor Antagonists Are Mediated by an Enhancement of GABAergic Synaptic Transmission via a Presynaptic CB1 Receptor Mechanism.Cells. 2022 Sep 27;11(19):3015. doi: 10.3390/cells11193015. Cells. 2022. PMID: 36230976 Free PMC article.
-
Progress and Pitfalls in Developing Agents to Treat Neurocognitive Deficits Associated with Schizophrenia.CNS Drugs. 2022 Aug;36(8):819-858. doi: 10.1007/s40263-022-00935-z. Epub 2022 Jul 13. CNS Drugs. 2022. PMID: 35831706 Free PMC article. Review.
-
Activating mGlu3 Metabotropic Glutamate Receptors Rescues Schizophrenia-like Cognitive Deficits Through Metaplastic Adaptations Within the Hippocampus.Biol Psychiatry. 2021 Sep 15;90(6):385-398. doi: 10.1016/j.biopsych.2021.02.970. Epub 2021 Mar 6. Biol Psychiatry. 2021. PMID: 33965197 Free PMC article.
-
Exception That Proves the Rule: Investigation of Privileged Stereochemistry in Designing Dopamine D3R Bitopic Agonists.ACS Med Chem Lett. 2020 Feb 28;11(10):1956-1964. doi: 10.1021/acsmedchemlett.9b00660. eCollection 2020 Oct 8. ACS Med Chem Lett. 2020. PMID: 33062179 Free PMC article.
-
Effects of the Positive Allosteric Modulator of Metabotropic Glutamate Receptor 5, VU-29, on Maintenance Association between Environmental Cues and Rewarding Properties of Ethanol in Rats.Biomolecules. 2020 May 20;10(5):793. doi: 10.3390/biom10050793. Biomolecules. 2020. PMID: 32443872 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
