Plasticity and genetic variation in traits underpinning asexual replication of the rodent malaria parasite, Plasmodium chabaudi
- PMID: 31262304
- PMCID: PMC6604315
- DOI: 10.1186/s12936-019-2857-0
Plasticity and genetic variation in traits underpinning asexual replication of the rodent malaria parasite, Plasmodium chabaudi
Abstract
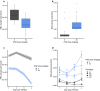
Background: The ability of malaria (Plasmodium) parasites to adjust investment into sexual transmission stages versus asexually replicating stages is well known, but plasticity in other traits underpinning the replication rate of asexual stages in the blood has received less attention. Such traits include burst size (the number of merozoites produced per schizont), the duration of the asexual cycle, and invasion preference for different ages of red blood cell (RBC).
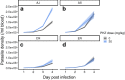
Methods: Here, plasticity [environment (E) effects] and genetic variation [genotype (G) effects] in traits relating to asexual replication rate are examined for 4 genotypes of the rodent malaria parasite Plasmodium chabaudi. An experiment tested whether asexual dynamics differ between parasites infecting control versus anaemic hosts, and whether variation in replication rate can be explained by differences in burst size, asexual cycle, and invasion rates.
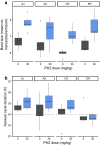
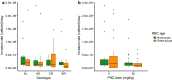
Results: The within-host environment affected each trait to different extents but generally had similar impacts across genotypes. The dynamics of asexual densities exhibited a genotype by environment effect (G×E), in which one of the genotypes increased replication rate more than the others in anaemic hosts. Burst size and cycle duration varied between the genotypes (G), while burst size increased and cycle duration became longer in anaemic hosts (E). Variation in invasion rates of differently aged RBCs was not explained by environmental or genetic effects. Plasticity in burst size and genotype are the only traits making significant contributions to the increase in asexual densities observed in anaemic hosts, together explaining 46.4% of the variation in replication rate.
Conclusions: That host anaemia induces several species of malaria parasites to alter conversion rate is well documented. Here, previously unknown plasticity in other traits underpinning asexual replication is revealed. These findings contribute to mounting evidence that malaria parasites deploy a suite of sophisticated strategies to maximize fitness by coping with, or exploiting the opportunities provided by, the variable within-host conditions experienced during infections. That genetic variation and genotype by environment interactions also shape these traits highlights their evolutionary potential. Asexual replication rate is a major determinant of virulence and so, understanding the evolution of virulence requires knowledge of the ecological (within-host environment) and genetic drivers of variation among parasites.
Keywords: Anaemia; Cycle duration; Genotype by environment; G×E; In-host survival; Life history trait; Phenotypic plasticity; Red blood cell preference; Virulence.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Adaptive plasticity in the gametocyte conversion rate of malaria parasites.PLoS Pathog. 2018 Nov 14;14(11):e1007371. doi: 10.1371/journal.ppat.1007371. eCollection 2018 Nov. PLoS Pathog. 2018. PMID: 30427935 Free PMC article.
-
Phenotypic plasticity in reproductive effort: malaria parasites respond to resource availability.Proc Biol Sci. 2017 Aug 16;284(1860):20171229. doi: 10.1098/rspb.2017.1229. Proc Biol Sci. 2017. PMID: 28768894 Free PMC article.
-
Information use and plasticity in the reproductive decisions of malaria parasites.Malar J. 2014 Mar 26;13:115. doi: 10.1186/1475-2875-13-115. Malar J. 2014. PMID: 24670151 Free PMC article.
-
The private life of malaria parasites: Strategies for sexual reproduction.Mol Biochem Parasitol. 2021 Jul;244:111375. doi: 10.1016/j.molbiopara.2021.111375. Epub 2021 May 20. Mol Biochem Parasitol. 2021. PMID: 34023299 Free PMC article. Review.
-
Virulence in malaria: an evolutionary viewpoint.Philos Trans R Soc Lond B Biol Sci. 2004 Jun 29;359(1446):965-86. doi: 10.1098/rstb.2003.1414. Philos Trans R Soc Lond B Biol Sci. 2004. PMID: 15306410 Free PMC article. Review.
Cited by
-
Periodic Parasites and Daily Host Rhythms.Cell Host Microbe. 2020 Feb 12;27(2):176-187. doi: 10.1016/j.chom.2020.01.005. Cell Host Microbe. 2020. PMID: 32053788 Free PMC article. Review.
-
Adaptive phenotypic plasticity in malaria parasites is not constrained by previous responses to environmental change.Evol Med Public Health. 2019 Sep 28;2019(1):190-198. doi: 10.1093/emph/eoz028. eCollection 2019. Evol Med Public Health. 2019. PMID: 31660151 Free PMC article.
-
Tritrichomonas foetus Cell Division Involves DNA Endoreplication and Multiple Fissions.Microbiol Spectr. 2023 Feb 2;11(2):e0325122. doi: 10.1128/spectrum.03251-22. Online ahead of print. Microbiol Spectr. 2023. PMID: 36728437 Free PMC article.
-
Challenges in forming inferences from limited data: a case study of malaria parasite maturation.J R Soc Interface. 2021 Apr;18(177):20210065. doi: 10.1098/rsif.2021.0065. Epub 2021 Apr 28. J R Soc Interface. 2021. PMID: 33906391 Free PMC article.
-
RNAscope in situ hybridization reveals microvascular sequestration of Plasmodium relictum pSGS1 blood stages but absence of exo-erythrocytic dormant stages during latent infection of Serinus canaria.Malar J. 2024 Mar 8;23(1):70. doi: 10.1186/s12936-024-04899-x. Malar J. 2024. PMID: 38459560 Free PMC article.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

