Mitochondrial protection by simvastatin against angiotensin II-mediated heart failure
- PMID: 31265743
- PMCID: PMC6780047
- DOI: 10.1111/bph.14781
Mitochondrial protection by simvastatin against angiotensin II-mediated heart failure
Erratum in
-
Correction.Br J Pharmacol. 2021 Sep;178(18):3845-3846. doi: 10.1111/bph.15624. Br J Pharmacol. 2021. PMID: 34431101 No abstract available.
Abstract
Background and purpose: Mitochondrial dysfunction plays a role in the progression of cardiovascular diseases including heart failure. 3-Hydroxy-3-methylglutaryl-CoA reductase inhibitors (statins), which inhibit ROS synthesis, show cardioprotective effects in chronic heart failure. However, the beneficial role of statins in mitochondrial protection in heart failure remains unclear.
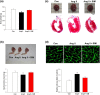
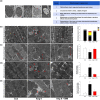
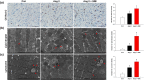
Experimental approach: Rats were treated with angiotensin II (1.5 mg·kg-1 ·day-1 ) or co-administered simvastatin (oral, 10 mg·kg-1 ) for 14 days; and then administration was stopped for the following 14 days. Cardiac structure/function was examined by wheat germ agglutinin staining and echocardiography. Mitochondrial morphology and the numbers of lipid droplets, lysosomes, autophagosomes, and mitophagosomes were determined by transmission electron microscopy. Human cardiomyocytes were stimulated, and intracellular ROS and mitochondrial membrane potential (ΔΨm ) changes were measured by flow cytometry and JC-1 staining, respectively. Autophagy and mitophagy-related and mitochondria-regulated apoptotic proteins were identified by immunohistochemistry and western blotting.
Key results: Simvastatin significantly reduced ROS production and attenuated the disruption of ΔΨm . Simvastatin induced the accumulation of lipid droplets to provide energy for maintaining mitochondrial function, promoted autophagy and mitophagy, and inhibited mitochondria-mediated apoptosis. These findings suggest that mitochondrial protection mediated by simvastatin plays a therapeutic role in heart failure prevention by modulating antioxidant status and promoting energy supplies for autophagy and mitophagy to inhibit mitochondrial damage and cardiomyocyte apoptosis.
Conclusion and implications: Mitochondria play a key role in mediating heart failure progression. Simvastatin attenuated heart failure, induced by angiotensin II, via mitochondrial protection and might provide a new therapy to prevent heart failure.
© 2019 The Authors. British Journal of Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Behavioral and quantitative mitochondrial proteome analyses of the effects of simvastatin: implications for models of neural degeneration.J Neural Transm (Vienna). 2009 Jul;116(7):791-806. doi: 10.1007/s00702-009-0247-4. Epub 2009 Jun 6. J Neural Transm (Vienna). 2009. PMID: 19504041
-
Simvastatin and rosuvastatin attenuate necroptosis in rat failing hearts following myocardial infarction; the contribution of Hsp90 inhibition in cardiomyocytes to prevent necroptosis.Biochem Pharmacol. 2025 Mar;233:116792. doi: 10.1016/j.bcp.2025.116792. Epub 2025 Jan 31. Biochem Pharmacol. 2025. PMID: 39894308
-
Simvastatin attenuates oxidant-induced mitochondrial dysfunction in cardiac myocytes.Circ Res. 2003 Oct 17;93(8):697-9. doi: 10.1161/01.RES.0000097262.21507.DF. Epub 2003 Sep 25. Circ Res. 2003. PMID: 14512440
-
[Statins and mitochondria].Postepy Biochem. 2016;62(2):77-84. Postepy Biochem. 2016. PMID: 28132458 Review. Polish.
-
Mitochondrial Stress as a Central Player in the Pathogenesis of Hypoxia-Related Myocardial Dysfunction: New Insights.Int J Med Sci. 2024 Sep 23;21(13):2502-2509. doi: 10.7150/ijms.99359. eCollection 2024. Int J Med Sci. 2024. PMID: 39439461 Free PMC article. Review.
Cited by
-
Lipid Droplet-a New Target in Ischemic Heart Disease.J Cardiovasc Transl Res. 2022 Aug;15(4):730-739. doi: 10.1007/s12265-021-10204-x. Epub 2022 Jan 4. J Cardiovasc Transl Res. 2022. PMID: 34984637 Review.
-
Reduced Immunity Regulator MAVS Contributes to Non-Hypertrophic Cardiac Dysfunction by Disturbing Energy Metabolism and Mitochondrial Homeostasis.Front Immunol. 2022 Jul 1;13:919038. doi: 10.3389/fimmu.2022.919038. eCollection 2022. Front Immunol. 2022. PMID: 35844503 Free PMC article.
-
Ablation of Shank3 alleviates cardiac dysfunction in aging mice by promoting CaMKII activation and Parkin-mediated mitophagy.Redox Biol. 2022 Dec;58:102537. doi: 10.1016/j.redox.2022.102537. Epub 2022 Nov 18. Redox Biol. 2022. PMID: 36436456 Free PMC article.
-
Simvastatin and fluvastatin attenuate trauma-induced cell death and catabolism in human cartilage.Front Bioeng Biotechnol. 2022 Sep 9;10:965302. doi: 10.3389/fbioe.2022.965302. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36159664 Free PMC article.
-
Breast Cancer and the Cardiovascular Disease: A Narrative Review.Cureus. 2022 Aug 12;14(8):e27917. doi: 10.7759/cureus.27917. eCollection 2022 Aug. Cureus. 2022. PMID: 36110451 Free PMC article. Review.
References
-
- Al‐Rasheed, N. M. , Al‐Oteibi, M. M. , Al‐Manee, R. Z. , Al‐Shareef, S. A. , Al‐Rasheed, N. M. , Hasan, I. H. , … Mahmoud, A. M. (2015). Simvastatin prevents isoproterenol‐induced cardiac hypertrophy through modulation of the JAK/STAT pathway. Drug Design, Development and Therapy, 9, 3217–3229. - PMC - PubMed
-
- Aubdool, A. A. , Thakore, P. , Argunhan, F. , Smillie, S. J. , Schnelle, M. , Srivastava, S. , … Brain, S. D. (2017). A novel α‐calcitonin gene‐related peptide analogue protects against end‐organ damage in experimental hypertension, cardiac hypertrophy, and heart failure. Circulation, 136, 367–383. 10.1161/CIRCULATIONAHA.117.028388 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

