Vagal mechanisms as neuromodulatory targets for the treatment of metabolic disease
- PMID: 31268181
- PMCID: PMC6810744
- DOI: 10.1111/nyas.14182
Vagal mechanisms as neuromodulatory targets for the treatment of metabolic disease
Abstract
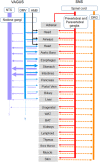
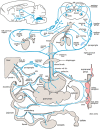
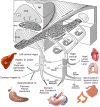
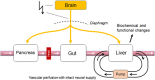
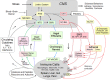
With few effective treatments available, the global rise of metabolic diseases, including obesity, type 2 diabetes mellitus, and cardiovascular disease, seems unstoppable. Likely caused by an obesogenic environment interacting with genetic susceptibility, the pathophysiology of obesity and metabolic diseases is highly complex and involves crosstalk between many organs and systems, including the brain. The vagus nerve is in a key position to bidirectionally link several peripheral metabolic organs with the brain and is increasingly targeted for neuromodulation therapy to treat metabolic disease. Here, we review the basics of vagal functional anatomy and its implications for vagal neuromodulation therapies. We find that most existing vagal neuromodulation techniques either ignore or misinterpret the rich functional specificity of both vagal efferents and afferents as demonstrated by a large body of literature. This lack of specificity of manipulating vagal fibers is likely the reason for the relatively poor beneficial long-term effects of such therapies. For these therapies to become more effective, rigorous validation of all physiological endpoints and optimization of stimulation parameters as well as electrode placements will be necessary. However, given the large number of function-specific fibers in any vagal branch, genetically guided neuromodulation techniques are more likely to succeed.
Keywords: anti-inflammatory pathways; diabetes; electrical stimulation; genetically guided; gut-brain communication; neuromodulation; obesity; vagotomy.
© 2019 The Authors. Annals of the New York Academy of Sciences published by Wiley Periodicals, Inc. on behalf of The New York Academy of Sciences.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

