Best Practices of Blood Cultures in Low- and Middle-Income Countries
- PMID: 31275940
- PMCID: PMC6591475
- DOI: 10.3389/fmed.2019.00131
Best Practices of Blood Cultures in Low- and Middle-Income Countries
Abstract
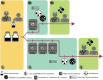
Bloodstream infections (BSI) have a substantial impact on morbidity and mortality worldwide. Despite scarcity of data from many low- and middle-income countries (LMICs), there is increasing awareness of the importance of BSI in these countries. For example, it is estimated that the global mortality of non-typhoidal Salmonella bloodstream infection in children under 5 already exceeds that of malaria. Reliable and accurate diagnosis of these infections is therefore of utmost importance. Blood cultures are the reference method for diagnosis of BSI. LMICs face many challenges when implementing blood cultures, due to financial, logistical, and infrastructure-related constraints. This review aims to provide an overview of the state-of-the-art of sampling and processing of blood cultures, with emphasis on its use in LMICs. Laboratory processing of blood cultures is relatively straightforward and can be done without the need for expensive and complicated equipment. Automates for incubation and growth monitoring have become the standard in high-income countries (HICs), but they are still too expensive and not sufficiently robust for imminent implementation in most LMICs. Therefore, this review focuses on "manual" methods of blood culture, not involving automated equipment. In manual blood cultures, a bottle consisting of a broth medium supporting bacterial growth is incubated in a normal incubator and inspected daily for signs of growth. The collection of blood for blood culture is a crucial step in the process, as the sensitivity of blood cultures depends on the volume sampled; furthermore, contamination of the blood culture (accidental inoculation of environmental and skin bacteria) can be avoided by appropriate antisepsis. In this review, we give recommendations regarding appropriate blood culture sampling and processing in LMICs. We present feasible methods to detect and speed up growth and discuss some challenges in implementing blood cultures in LMICs, such as the biosafety aspects, supply chain and waste management.
Keywords: bacteremia diagnosis; blood culture; clinical bacteriology; laboratory medicine practices; low-resource settings (LRS).
Figures







References
-
- Baron E, Dunne W, Weinstein M, Weich D, Wilson D, Yagupsky P. Cumitech 1C, Blood cultures IV. In: Baron E, editor. Cumitech 1C. Washington, DC: ASM Press; (2005), 1–34.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

