Elevations in Circulating sST2 Levels Are Associated With In-Hospital Mortality and Adverse Clinical Outcomes After Blunt Trauma
- PMID: 31279260
- PMCID: PMC6815699
- DOI: 10.1016/j.jss.2019.05.057
Elevations in Circulating sST2 Levels Are Associated With In-Hospital Mortality and Adverse Clinical Outcomes After Blunt Trauma
Abstract
Background: Soluble suppression of tumorigenicity 2 (sST2), a decoy receptor for interleukin (IL)-33, has emerged as a novel biomarker in various disease processes. Recent studies have elucidated the role of the sST2/IL-33 complex in modulating the balance of Th1/Th2 immune responses after tissue stress. However, the role of sST2 as a biomarker after traumatic injury remains unclear. To address this, we evaluated serum sST2 correlations with mortality and in-hospital adverse outcomes as endpoints in blunt trauma patients.
Methods: We retrospectively analyzed clinical and biobank data of 493 blunt trauma victims 472 survivors (mean age: 48.4 ± 0.87; injury severity score [ISS]: 19.6 ± 0.48) and 19 nonsurvivors (mean age: 58.8 ± 4.5; ISS: 23.3 ± 2.1) admitted to the intensive care unit. Given the confounding impact of age on the inflammatory response, we derived a propensity-matched survivor subgroup (n = 19; mean age: 59 ± 3; ISS: 23.4 ± 2) using an IBM SPSS case-control matching algorithm. Serial blood samples were obtained from all patients (3 samples within the first 24 h and then once daily from day [D] 1 to D5 after injury). sST2 and twenty-nine inflammatory biomarkers were assayed using enzyme-linked immunosorbent assay and Luminex, respectively. Two-way analysis of variance on ranks was used to compare groups (P < 0.05). Spearman rank correlation was performed to determine the association of circulating sST2 levels with biomarker levels and in-hospital clinical outcomes.
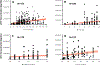
Results: Circulating sST2 levels of the nonsurvivor cohort were statistically significantly elevated at 12 h after injury and remained elevated up to D5 when compared either to the overall 472 survivor cohort or a matched 19 survivor subcohort. Admission sST2 levels obtained from the first blood draw after injury in the survivor cohort correlated positively with admission base deficit (correlation coefficient [CC] = 0.1; P = 0.02), international normalized ratio (CC = 0.1, P = 0.03), ISS (CC = 0.1, P = 0.008), and the average Marshall multiple organ dysfunction score between D2 and D5 (CC = 0.1, P = 0.04). Correlations with ISS revealed a positive correlation of ISS with plasma sST2 levels across the mild ISS (CC = 0.47, P < 0.001), moderate ISS (CC = 0.58, P < 0.001), and severe ISS groups (CC = 0.63, P < 0.001). Analysis of biomarker correlations in the matched survivor group over the initial 24 h after injury showed that sST2 correlates strongly and positively with IL-4 (CC = 0.65, P = 0.002), IL-5 (CC = 0.57, P = 0.01), IL-21 (CC = 0.52, P = 0.02), IL-2 (CC = 0.51, P = 0.02), soluble IL-2 receptor-α (CC = 0.5, P = 0.02), IL-13 (CC = 0.49, P = 0.02), and IL-17A (CC = 0.48, P = 0.03). This was not seen in the matched nonsurvivor group. sST2/IL-33 ratios were significantly elevated in nonsurvivors and patients with severe injury based on ISS ≥ 25.
Conclusions: Elevations in serum sST2 levels are associated with poor clinical trajectories and mortality after blunt trauma. High sST2 coupled with low IL-33 associates with severe injury, mortality, and worse clinical outcomes. These findings suggest that sST2 could serve as an early prognostic biomarker in trauma patients and that sustained elevations of sST2 could contribute to a detrimental suppression of IL-33 bioavailability in patients with high injury severity.
Keywords: Acute inflammation; Base deficit; Blunt trauma; Clinical outcomes; Coagulopathy; Inflammatory biomarkers; Injury severity; Intensive care unit; Multiple organ dysfunction syndrome; Soluble suppression of tumorigenicity.
Copyright © 2019 Elsevier Inc. All rights reserved.
Figures
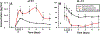
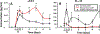
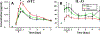
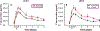
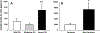
Similar articles
-
Impact of Injury Severity on Dynamic Inflammation Networks Following Blunt Trauma.Shock. 2015 Aug;44(2):101-9. doi: 10.1097/SHK.0000000000000395. Shock. 2015. PMID: 26009819 Free PMC article.
-
Diurnal Variation in Systemic Acute Inflammation and Clinical Outcomes Following Severe Blunt Trauma.Front Immunol. 2019 Nov 20;10:2699. doi: 10.3389/fimmu.2019.02699. eCollection 2019. Front Immunol. 2019. PMID: 31824494 Free PMC article.
-
IL33-mediated ILC2 activation and neutrophil IL5 production in the lung response after severe trauma: A reverse translation study from a human cohort to a mouse trauma model.PLoS Med. 2017 Jul 25;14(7):e1002365. doi: 10.1371/journal.pmed.1002365. eCollection 2017 Jul. PLoS Med. 2017. PMID: 28742815 Free PMC article.
-
The clinicopathological implications of serum IL-33 and sST2 as cancer biomarkers: A narrative review.J Oral Biol Craniofac Res. 2025 May-Jun;15(3):645-658. doi: 10.1016/j.jobcr.2025.03.016. Epub 2025 Apr 10. J Oral Biol Craniofac Res. 2025. PMID: 40271080 Free PMC article. Review.
-
High-dimensional proteomics identifies organ injury patterns associated with outcomes in human trauma.J Trauma Acute Care Surg. 2023 Jun 1;94(6):803-813. doi: 10.1097/TA.0000000000003880. Epub 2023 Feb 13. J Trauma Acute Care Surg. 2023. PMID: 36787435 Free PMC article. Review.
Cited by
-
Protective/reparative cytokines are suppressed at high injury severity in human trauma.Trauma Surg Acute Care Open. 2021 Mar 2;6(1):e000619. doi: 10.1136/tsaco-2020-000619. eCollection 2021. Trauma Surg Acute Care Open. 2021. PMID: 33748428 Free PMC article.
-
Prehospital plasma is associated with distinct biomarker expression following injury.JCI Insight. 2020 Apr 23;5(8):e135350. doi: 10.1172/jci.insight.135350. JCI Insight. 2020. PMID: 32229722 Free PMC article. Clinical Trial.
-
Serum soluble suppression of tumorigenicity 2 as a novel inflammatory marker predicts the severity of acute pancreatitis.World J Gastroenterol. 2021 Oct 14;27(38):6489-6500. doi: 10.3748/wjg.v27.i38.6489. World J Gastroenterol. 2021. PMID: 34720537 Free PMC article.
-
Analysis of the Plasma Metabolome after Trauma, Novel Circulating Sphingolipid Signatures, and In-Hospital Outcomes.J Am Coll Surg. 2021 Mar;232(3):276-287.e1. doi: 10.1016/j.jamcollsurg.2020.12.022. Epub 2021 Jan 14. J Am Coll Surg. 2021. PMID: 33453380 Free PMC article. Clinical Trial.
-
The Use of Multiplexing to Identify Cytokine and Chemokine Networks in the Immune-Inflammatory Response to Trauma.Antioxid Redox Signal. 2021 Dec;35(16):1393-1406. doi: 10.1089/ars.2021.0054. Epub 2021 May 19. Antioxid Redox Signal. 2021. PMID: 33860683 Free PMC article. Review.
References
-
- Namas RA, Vodovotz Y, Almahmoud K, Abdul-Malak O, Zaaqoq A, Namas R, Mi Q, Barclay D, Zuckerbraun B, Peitzman AB et al.: Temporal Patterns of Circulating Inflammation Biomarker Networks Differentiate Susceptibility to Nosocomial Infection Following Blunt Trauma in Humans. Ann Surg 2016, 263(1):191–198. - PMC - PubMed
-
- Tschoeke SK, Ertel W: Immunoparalysis after multiple trauma. Injury 2007, 38(12):1346–1357. - PubMed
-
- Namas RA, Mi Q, Namas R, Almahmoud K, Zaaqoq AM, Abdul-Malak O, Azhar N, Day J, Abboud A, Zamora R et al.: Insights into the Role of Chemokines, Damage-Associated Molecular Patterns, and Lymphocyte-Derived Mediators from Computational Models of Trauma-Induced Inflammation. Antioxidants & redox signaling 2015, 23(17):1370–1387. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

