Genomic Delineation of Zoonotic Origins of Clostridium difficile
- PMID: 31281807
- PMCID: PMC6595230
- DOI: 10.3389/fpubh.2019.00164
Genomic Delineation of Zoonotic Origins of Clostridium difficile
Abstract
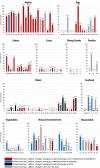
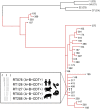
Clostridium difficile is toxin-producing antimicrobial resistant (AMR) enteropathogen historically associated with diarrhea and pseudomembranous colitis in hospitalized patients. In recent years, there have been dramatic increases in the incidence and severity of C. difficile infection (CDI), and associated morbidity and mortality, in both healthcare and community settings. C. difficile is an ancient and diverse species that displays a sympatric lifestyle, establishing itself in a range of ecological niches external to the healthcare system. These sources/reservoirs include food, water, soil, and over a dozen animal species, in particular, livestock such as pigs and cattle. In a manner analogous to human infection, excessive antimicrobial exposure, particularly to cephalosporins, is driving the expansion of C. difficile in livestock populations worldwide. Subsequent spore contamination of meat, vegetables grown in soil containing animal feces, agricultural by-products such as compost and manure, and the environment in general (households, lawns, and public spaces) is contributing to a persistent community source/reservoir of C. difficile and the insidious rise of CDI in the community. The whole-genome sequencing era continues to redefine our view of this complex pathogen. The application of high-resolution microbial genomics in a One Health framework (encompassing clinical, veterinary, and environment derived datasets) is the optimal paradigm for advancing our understanding of CDI in humans and animals. This approach has begun to yield critical insights into the genetic diversity, evolution, AMR, and zoonotic potential of C. difficile. In Europe, North America, and Australia, microevolutionary analysis of the C. difficile core genome shows strains common to humans and animals (livestock or companion animals) do not form distinct populations but share a recent evolutionary history. Moreover, for C. difficile sequence type 11 and PCR ribotypes 078 and 014, major lineages of One Health importance, this approach has substantiated inter-species clonal transmission between animals and humans. These findings indicate either a zoonosis or anthroponosis. Moreover, they challenge the existing paradigm and the long-held misconception that CDI is primarily a healthcare-associated infection. In this article, evolutionary, and zoonotic aspects of CDI are discussed, including the anthropomorphic factors that contribute to the spread of C. difficile from the farm to the community.
Keywords: Clostridium difficile; evolution; livestock; one health; transmission; zoonosis.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources

