Evolution of the Chordate Telencephalon
- PMID: 31287987
- PMCID: PMC11073819
- DOI: 10.1016/j.cub.2019.05.026
Evolution of the Chordate Telencephalon
Abstract
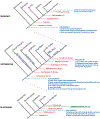
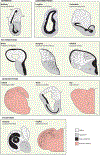
The dramatic evolutionary expansion of the neocortex, together with a proliferation of specialized cortical areas, is believed to underlie the emergence of human cognitive abilities. In a broader phylogenetic context, however, neocortex evolution in mammals, including humans, is remarkably conservative, characterized largely by size variations on a shared six-layered neuronal architecture. By contrast, the telencephalon in non-mammalian vertebrates, including reptiles, amphibians, bony and cartilaginous fishes, and cyclostomes, features a great variety of very different tissue structures. Our understanding of the evolutionary relationships of these telencephalic structures, especially those of basally branching vertebrates and invertebrate chordates, remains fragmentary and is impeded by conceptual obstacles. To make sense of highly divergent anatomies requires a hierarchical view of biological organization, one that permits the recognition of homologies at multiple levels beyond neuroanatomical structure. Here we review the origin and diversification of the telencephalon with a focus on key evolutionary innovations shaping the neocortex at multiple levels of organization.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Figures


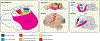
References
-
- Gee H (2018). Across the Bridge: Understanding the Origin of the Vertebrates (Chicago: The University of Chicago Press; ).
-
- Erwin DH, and Valentine JW (2013). The Cambrian Explosion: The Construction of Animal Biodiversity (Greenwood Village, Colorado: Roberts and Company; ).
-
- Gans C, and Northcutt RG (1983). Neural crest and the origin of vertebrates: a new head. Science 220, 268–273. - PubMed
-
- Nieuwenhuys R, Donkelaar H.J.t., and Nicholson C (1998). The Central Nervous System of Vertebrates (New York: Springer; ).
-
- Butler AB, and Hodos W (2005). Comparative Vertebrate Neuroanatomy: Evolution and Adaptation, 2nd Edition. (Hoboken, NJ: Wiley-Interscience; ).
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

