Manganese-Induced Neurotoxicity: New Insights Into the Triad of Protein Misfolding, Mitochondrial Impairment, and Neuroinflammation
- PMID: 31293375
- PMCID: PMC6606738
- DOI: 10.3389/fnins.2019.00654
Manganese-Induced Neurotoxicity: New Insights Into the Triad of Protein Misfolding, Mitochondrial Impairment, and Neuroinflammation
Abstract
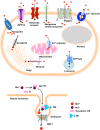
Occupational or environmental exposure to manganese (Mn) can lead to the development of "Manganism," a neurological condition showing certain motor symptoms similar to Parkinson's disease (PD). Like PD, Mn toxicity is seen in the central nervous system mainly affecting nigrostriatal neuronal circuitry and subsequent behavioral and motor impairments. Since the first report of Mn-induced toxicity in 1837, various experimental and epidemiological studies have been conducted to understand this disorder. While early investigations focused on the impact of high concentrations of Mn on the mitochondria and subsequent oxidative stress, current studies have attempted to elucidate the cellular and molecular pathways involved in Mn toxicity. In fact, recent reports suggest the involvement of Mn in the misfolding of proteins such as α-synuclein and amyloid, thus providing credence to the theory that environmental exposure to toxicants can either initiate or propagate neurodegenerative processes by interfering with disease-specific proteins. Besides manganism and PD, Mn has also been implicated in other neurological diseases such as Huntington's and prion diseases. While many reviews have focused on Mn homeostasis, the aim of this review is to concisely synthesize what we know about its effect primarily on the nervous system with respect to its role in protein misfolding, mitochondrial dysfunction, and consequently, neuroinflammation and neurodegeneration. Based on the current evidence, we propose a 'Mn Mechanistic Neurotoxic Triad' comprising (1) mitochondrial dysfunction and oxidative stress, (2) protein trafficking and misfolding, and (3) neuroinflammation.
Keywords: Parkinson’s disease; cell-to-cell transmission and neuroinflammation; exosome; manganese neurotoxicity; protein aggregation.
Figures




References
-
- Afeseh Ngwa H., Kanthasamy A., Anantharam V., Song C., Witte T., Houk R., et al. (2009). Vanadium induces dopaminergic neurotoxicity via protein kinase C-delta dependent oxidative signaling mechanisms: relevance to etiopathogenesis of Parkinson’s disease. Toxicol. Appl. Pharmacol. 240 273–285. 10.1016/j.taap.2009.07.025 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

