Combined Cyclosporin A and Hypothermia Treatment Inhibits Activation of BV-2 Microglia but Induces an Inflammatory Response in an Ischemia/Reperfusion Hippocampal Slice Culture Model
- PMID: 31293389
- PMCID: PMC6603137
- DOI: 10.3389/fncel.2019.00273
Combined Cyclosporin A and Hypothermia Treatment Inhibits Activation of BV-2 Microglia but Induces an Inflammatory Response in an Ischemia/Reperfusion Hippocampal Slice Culture Model
Abstract
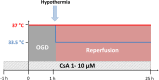
Introduction: Hypothermia attenuates cerebral ischemia-induced neuronal cell death associated with neuroinflammation. The calcineurin inhibitor cyclosporin A (CsA) has been shown to be neuroprotective by minimizing activation of inflammatory pathways. Therefore, we investigated whether the combination of hypothermia and treatment with CsA has neuroprotective effects in an oxygen-glucose deprivation/reperfusion (OGD/R) injury model in neuronal and BV-2 microglia monocultures, as well as in an organotypic hippocampal slice culture (OHSC).
Methods: Murine primary neurons, BV-2 microglia, and OHSC were pretreated with CsA and exposed to 1 h OGD (0.2% O2) followed by reperfusion at normothermia (37°C) or hypothermia (33.5°C). Cytotoxicity was measured by lactate dehydrogenase and glutamate releases. Damage-associated molecular patterns (DAMPs) high mobility group box 1 (HMGB1), heat shock protein 70 (Hsp70), and cold-inducible RNA-binding protein (CIRBP) were detected in cultured supernatant by western blot analysis. Interleukin-6 (IL-6), Interleukin-1α and -1β (IL-1α/IL1-β), tumor necrosis factor-α (TNF-α), monocyte chemotactic protein 1 (MCP1), inducible nitric oxide synthase (iNOS), glia activation factors ionized calcium-binding adapter molecule 1 (Iba1), and transforming growth factor β1 (TGF-β1) gene expressions were analyzed by RT-qPCR.
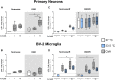
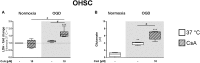
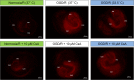
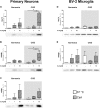
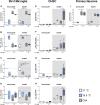
Results: Exposure to OGD plus 10 μM CsA was sufficient to induce necrotic cell death and subsequent release of DAMPs in neurons but not BV-2 microglia. Moreover, OGD/R-induced secondary injury was also observed only in the neurons, which was not attenuated by cooling and no increased toxicity by CsA was observed. BV-2 microglia were not sensitive to OGD/R-induced injury but were susceptible to CsA-induced toxicity in a dose dependent manner, which was minimized by hypothermia. CsA attenuated IL-1β and Iba1 expressions in BV-2 microglia exposed to OGD/R. Hypothermia reduced IL-1β and iNOS expressions but induced TNF-α and Iba1 expressions in the microglia. However, these observations did not translate to the ex vivo OHCS model, as general high expressions of most cytokines investigated were observed.
Conclusion: Treatment with CsA has neurotoxic effects on primary neurons exposed to OGD but could inhibit BV-2 microglia activation. However, CsA and hypothermia treatment after ischemia/reperfusion injury results in cytotoxic neuroinflammation in the complex ex vivo OHSC.
Keywords: BV-2 microglia; DAMPs; cyclosporin A; hypothermia; inflammation; organotypic hippocampal slice culture; oxygen-glucose deprivation/reperfusion; primary neuron.
Figures

Similar articles
-
Cooling and Sterile Inflammation in an Oxygen-Glucose-Deprivation/Reperfusion Injury Model in BV-2 Microglia.Mediators Inflamm. 2021 Nov 5;2021:8906561. doi: 10.1155/2021/8906561. eCollection 2021. Mediators Inflamm. 2021. PMID: 34776788 Free PMC article.
-
Hypothermia-Induced Ubiquitination of Voltage-Dependent Anion Channel 3 Protects BV2 Microglia Cells From Cytotoxicity Following Oxygen-Glucose Deprivation/Recovery.Front Mol Neurosci. 2020 Jun 5;13:100. doi: 10.3389/fnmol.2020.00100. eCollection 2020. Front Mol Neurosci. 2020. PMID: 32581711 Free PMC article.
-
The microRNA miR-181c controls microglia-mediated neuronal apoptosis by suppressing tumor necrosis factor.J Neuroinflammation. 2012 Sep 6;9:211. doi: 10.1186/1742-2094-9-211. J Neuroinflammation. 2012. PMID: 22950459 Free PMC article.
-
Effects of moderate and deep hypothermia on RNA-binding proteins RBM3 and CIRP expressions in murine hippocampal brain slices.Brain Res. 2013 Apr 4;1504:74-84. doi: 10.1016/j.brainres.2013.01.041. Epub 2013 Feb 8. Brain Res. 2013. PMID: 23415676
-
Experimental Models for Testing the Efficacy of Pharmacological Treatments for Neonatal Hypoxic-Ischemic Encephalopathy.Biomedicines. 2022 Apr 19;10(5):937. doi: 10.3390/biomedicines10050937. Biomedicines. 2022. PMID: 35625674 Free PMC article. Review.
Cited by
-
Extremely Low-Frequency Electromagnetic Stimulation (ELF-EMS) Improves Neurological Outcome and Reduces Microglial Reactivity in a Rodent Model of Global Transient Stroke.Int J Mol Sci. 2023 Jul 5;24(13):11117. doi: 10.3390/ijms241311117. Int J Mol Sci. 2023. PMID: 37446295 Free PMC article.
-
Mild Therapeutic Hypothermia Protects from Acute and Chronic Renal Ischemia-Reperfusion Injury in Mice by Mitigated Mitochondrial Dysfunction and Modulation of Local and Systemic Inflammation.Int J Mol Sci. 2022 Aug 17;23(16):9229. doi: 10.3390/ijms23169229. Int J Mol Sci. 2022. PMID: 36012493 Free PMC article.
-
Bone Marrow-Derived Mononuclear Cell Transplants Decrease Retinal Gliosis in Two Animal Models of Inherited Photoreceptor Degeneration.Int J Mol Sci. 2020 Sep 30;21(19):7252. doi: 10.3390/ijms21197252. Int J Mol Sci. 2020. PMID: 33008136 Free PMC article.
-
Cooling and Sterile Inflammation in an Oxygen-Glucose-Deprivation/Reperfusion Injury Model in BV-2 Microglia.Mediators Inflamm. 2021 Nov 5;2021:8906561. doi: 10.1155/2021/8906561. eCollection 2021. Mediators Inflamm. 2021. PMID: 34776788 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials

