Canine IL4-10 fusion protein provides disease modifying activity in a canine model of OA; an exploratory study
- PMID: 31295306
- PMCID: PMC6622543
- DOI: 10.1371/journal.pone.0219587
Canine IL4-10 fusion protein provides disease modifying activity in a canine model of OA; an exploratory study
Abstract
Objective: An ideal disease modifying osteoarthritis drug (DMOAD) has chondroprotective, anti-inflammatory, and analgesic effects. This study describes the production and characterization of a canine IL4-10 fusion protein (IL4-10 FP) and evaluates its in vivo DMOAD activity in a canine model of osteoarthritis (OA).
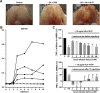
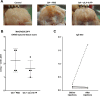
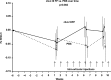
Design: The canine Groove model was used as an in vivo model of degenerative knee OA. Six weeks after OA induction dogs were intra-articularly injected weekly, for ten weeks, with either IL4-10 FP or phosphate buffered saline (PBS). In addition to the use of human IL4-10 FP, canine IL4-10 FP was developed and characterized in vitro, and tested in vivo. Force plate analysis (FPA) was performed to analyze joint loading as a proxy measure for pain. After ten weeks dogs were euthanized and cartilage and synovial tissue samples were analyzed by histochemistry (OARSI scores) and biochemistry (cartilage proteoglycan turnover).
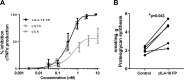
Results: Repetitive intra-articular injections with human IL4-10 FP led to antibody formation, that blocked its functional activity. Therefore, a canine IL4-10 FP was developed, which completely inhibited LPS-induced TNFα production by canine blood cells, and increased proteoglycan synthesis of canine cartilage in vitro (p = 0.043). In vivo, canine IL4-10 FP restored the, by OA impaired, joint loading (p = 0.002) and increased cartilage proteoglycan content (p = 0.029).
Conclusions: This first study on the potential DMOAD activity upon prolonged repeated treatment with IL4-10 FP demonstrates that a species-specific variant has anti-inflammatory and chondroprotective effects in vitro and chondroprotective and analgesic effects in vivo. These data warrant further research on the DMOAD potential of the IL4-10 FP.
Conflict of interest statement
FL and CEH are inventors on the patent WO2013070076 owned by UMC Utrecht Holdings. This does not alter our adherence to PLOS ONE policies on sharing data and materials. JP, NE, and CEH are shareholders of Synerkine Pharma BV, a spin-off of the UMC Utrecht licensing the patents on the IL4-10 fusion protein.
Figures




Similar articles
-
Candidates for Intra-Articular Administration Therapeutics and Therapies of Osteoarthritis.Int J Mol Sci. 2021 Mar 30;22(7):3594. doi: 10.3390/ijms22073594. Int J Mol Sci. 2021. PMID: 33808364 Free PMC article. Review.
-
IL4-10 fusion protein has chondroprotective, anti-inflammatory and potentially analgesic effects in the treatment of osteoarthritis.Osteoarthritis Cartilage. 2018 Aug;26(8):1127-1135. doi: 10.1016/j.joca.2018.05.005. Epub 2018 May 26. Osteoarthritis Cartilage. 2018. PMID: 29775732
-
IL4-10 Fusion Protein Shows DMOAD Activity in a Rat Osteoarthritis Model.Cartilage. 2021 Dec;13(2_suppl):1155S-1164S. doi: 10.1177/19476035211026736. Epub 2021 Jun 23. Cartilage. 2021. PMID: 34159843 Free PMC article.
-
The Role of Interleukin-4 and Interleukin-10 in Osteoarthritic Joint Disease: A Systematic Narrative Review.Cartilage. 2022 Apr-Jun;13(2):19476035221098167. doi: 10.1177/19476035221098167. Cartilage. 2022. PMID: 35549461 Free PMC article.
-
IL4-10 fusion protein: a novel immunoregulatory drug combining activities of interleukin 4 and interleukin 10.Clin Exp Immunol. 2019 Jan;195(1):1-9. doi: 10.1111/cei.13224. Epub 2018 Nov 11. Clin Exp Immunol. 2019. PMID: 30307604 Free PMC article.
Cited by
-
Exploring Intra-Articular Administration of Monoclonal Antibodies as a Novel Approach to Osteoarthritis Treatment: A Systematic Review.Biomedicines. 2024 Sep 28;12(10):2217. doi: 10.3390/biomedicines12102217. Biomedicines. 2024. PMID: 39457530 Free PMC article. Review.
-
Enhanced Extracellular Matrix Breakdown Characterizes the Early Distraction Phase of Canine Knee Joint Distraction.Cartilage. 2021 Dec;13(2_suppl):1654S-1664S. doi: 10.1177/19476035211014595. Epub 2021 May 20. Cartilage. 2021. PMID: 34014119 Free PMC article.
-
Candidates for Intra-Articular Administration Therapeutics and Therapies of Osteoarthritis.Int J Mol Sci. 2021 Mar 30;22(7):3594. doi: 10.3390/ijms22073594. Int J Mol Sci. 2021. PMID: 33808364 Free PMC article. Review.
-
Dorsal Root Ganglia Macrophages Maintain Osteoarthritis Pain.J Neurosci. 2021 Sep 29;41(39):8249-8261. doi: 10.1523/JNEUROSCI.1787-20.2021. Epub 2021 Aug 16. J Neurosci. 2021. PMID: 34400519 Free PMC article.
-
Experimental Therapeutics for the Treatment of Osteoarthritis.J Exp Pharmacol. 2021 Feb 11;13:101-125. doi: 10.2147/JEP.S237479. eCollection 2021. J Exp Pharmacol. 2021. PMID: 33603501 Free PMC article. Review.
References
-
- Bijlsma JW, Berenbaum F, Lafeber FP. Osteoarthritis: an update with relevance for clinical practice. Lancet (London, England). 2011;377(9783):2115–26. - PubMed
-
- Moore EE, Bendele AM, Thompson DL, Littau A, Waggie KS, Reardon B, et al. Fibroblast growth factor-18 stimulates chondrogenesis and cartilage repair in a rat model of injury-induced osteoarthritis. Osteoarthritis and cartilage / OARS, Osteoarthritis Research Society. 2005;13(7):623–31. - PubMed
-
- Lohmander LS, Hellot S, Dreher D, Krantz EF, Kruger DS, Guermazi A, et al. Intraarticular sprifermin (recombinant human fibroblast growth factor 18) in knee osteoarthritis: a randomized, double-blind, placebo-controlled trial. Arthritis & rheumatology (Hoboken, NJ). 2014;66(7):1820–31. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

