Digital health: a path to validation
- PMID: 31304384
- PMCID: PMC6550273
- DOI: 10.1038/s41746-019-0111-3
Digital health: a path to validation
Abstract
Digital health solutions continue to grow in both number and capabilities. Despite these advances, the confidence of the various stakeholders - from patients and clinicians to payers, industry and regulators - in medicine remains quite low. As a result, there is a need for objective, transparent, and standards-based evaluation of digital health products that can bring greater clarity to the digital health marketplace. We believe an approach that is guided by end-user requirements and formal assessment across technical, clinical, usability, and cost domains is one possible solution. For digital health solutions to have greater impact, quality and value must be easier to distinguish. To that end, we review the existing landscape and gaps, highlight the evolving responses and approaches, and detail one pragmatic framework that addresses the current limitations in the marketplace with a path toward implementation.
Keywords: Health policy; Technology.
Conflict of interest statement
Competing interestsThe authors declare no competing interests.
Figures
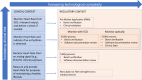
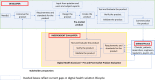

References
-
- Rock Health. 2016 Year End Funding Report: A Reality Check for Digital Health. (San Francisco, CA, Rock Health, 2017). https://rockhealth.com/reports/2016-year-end-funding-report-a-reality-ch....
-
- World Health Organization. Classification of digital health interventions v1. 0: a shared language to describe the uses of digital technology for health. World Health Organization (2018).
-
- Rock Health. 2017 Year End Funding Report: The End of the Beginning of Digital Health. https://rockhealth.com/reports/2017-year-end-funding-report-the-end-of-t... (2018).
-
- IQVIA. IQVIA Institute for Human Data Science Study: Impact of Digital Health Grows as Innovation, Evidence and Adoption of Mobile Health Apps Accelerate.https://www.iqvia.com/newsroom/2017/11/impact-of-digital-health-grows-as... (2017).
Publication types
LinkOut - more resources
Full Text Sources

