Impaired skeletal muscle mitochondrial pyruvate uptake rewires glucose metabolism to drive whole-body leanness
- PMID: 31305240
- PMCID: PMC6684275
- DOI: 10.7554/eLife.45873
Impaired skeletal muscle mitochondrial pyruvate uptake rewires glucose metabolism to drive whole-body leanness
Abstract
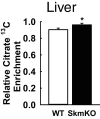
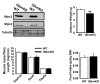
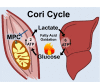
Metabolic cycles are a fundamental element of cellular and organismal function. Among the most critical in higher organisms is the Cori Cycle, the systemic cycling between lactate and glucose. Here, skeletal muscle-specific Mitochondrial Pyruvate Carrier (MPC) deletion in mice diverted pyruvate into circulating lactate. This switch disinhibited muscle fatty acid oxidation and drove Cori Cycling that contributed to increased energy expenditure. Loss of muscle MPC activity led to strikingly decreased adiposity with complete muscle mass and strength retention. Notably, despite decreasing muscle glucose oxidation, muscle MPC disruption increased muscle glucose uptake and whole-body insulin sensitivity. Furthermore, chronic and acute muscle MPC deletion accelerated fat mass loss on a normal diet after high fat diet-induced obesity. Our results illuminate the role of the skeletal muscle MPC as a whole-body carbon flux control point. They highlight the potential utility of modulating muscle pyruvate utilization to ameliorate obesity and type 2 diabetes.
Keywords: cell biology; cori cycle; diabetes; glucose uptake; human biology; medicine; mitochondrial pyruvate carrier; mouse; obesity; skeletal muscle.
© 2019, Sharma et al.
Conflict of interest statement
AS, LO, RS, AR, ZZ, ST, KC, WG, LG, DS, AP, EC, MD, JC, CA, BD, RS, AN, GP, LZ, ET No competing interests declared
Figures










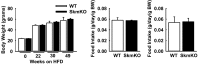
Similar articles
-
The mitochondrial pyruvate carrier mediates high fat diet-induced increases in hepatic TCA cycle capacity.Mol Metab. 2017 Nov;6(11):1468-1479. doi: 10.1016/j.molmet.2017.09.002. Epub 2017 Sep 18. Mol Metab. 2017. PMID: 29107293 Free PMC article.
-
Embryonic Lethality of Mitochondrial Pyruvate Carrier 1 Deficient Mouse Can Be Rescued by a Ketogenic Diet.PLoS Genet. 2016 May 13;12(5):e1006056. doi: 10.1371/journal.pgen.1006056. eCollection 2016 May. PLoS Genet. 2016. PMID: 27176894 Free PMC article.
-
Mitochondrial pyruvate carrier inhibitors improve metabolic parameters in diet-induced obese mice.J Biol Chem. 2022 Feb;298(2):101554. doi: 10.1016/j.jbc.2021.101554. Epub 2021 Dec 30. J Biol Chem. 2022. PMID: 34973337 Free PMC article.
-
The mitochondrial pyruvate carrier in health and disease: To carry or not to carry?Biochim Biophys Acta. 2016 Oct;1863(10):2436-42. doi: 10.1016/j.bbamcr.2016.01.017. Epub 2016 Jan 26. Biochim Biophys Acta. 2016. PMID: 26826034 Review.
-
Mitochondrial pyruvate import and its effects on homeostasis.Curr Opin Cell Biol. 2015 Apr;33:35-41. doi: 10.1016/j.ceb.2014.10.008. Epub 2014 Nov 15. Curr Opin Cell Biol. 2015. PMID: 25463844 Review.
Cited by
-
The mitochondrial lactate oxidation complex: endpoint for carbohydrate carbon disposal.Am J Physiol Endocrinol Metab. 2025 Jan 1;328(1):E126-E136. doi: 10.1152/ajpendo.00306.2024. Epub 2024 Dec 23. Am J Physiol Endocrinol Metab. 2025. PMID: 39714986 Free PMC article.
-
Mitochondrial Pyruvate Carrier Function in Health and Disease across the Lifespan.Biomolecules. 2020 Aug 8;10(8):1162. doi: 10.3390/biom10081162. Biomolecules. 2020. PMID: 32784379 Free PMC article. Review.
-
Profile of gut microbiota and serum metabolites associated with metabolic syndrome in a remote island most afflicted by obesity in Japan.Sci Rep. 2022 Oct 14;12(1):17292. doi: 10.1038/s41598-022-21708-0. Sci Rep. 2022. PMID: 36241691 Free PMC article.
-
Energy metabolism in health and diseases.Signal Transduct Target Ther. 2025 Feb 18;10(1):69. doi: 10.1038/s41392-025-02141-x. Signal Transduct Target Ther. 2025. PMID: 39966374 Free PMC article. Review.
-
The Importance of Mitochondrial Pyruvate Carrier in Cancer Cell Metabolism and Tumorigenesis.Cancers (Basel). 2021 Mar 24;13(7):1488. doi: 10.3390/cancers13071488. Cancers (Basel). 2021. PMID: 33804985 Free PMC article. Review.
References
-
- Bartelt A, Bruns OT, Reimer R, Hohenberg H, Ittrich H, Peldschus K, Kaul MG, Tromsdorf UI, Weller H, Waurisch C, Eychmüller A, Gordts PL, Rinninger F, Bruegelmann K, Freund B, Nielsen P, Merkel M, Heeren J. Brown adipose tissue activity controls triglyceride clearance. Nature Medicine. 2011;17:200–205. doi: 10.1038/nm.2297. - DOI - PubMed
-
- Bricker DK, Taylor EB, Schell JC, Orsak T, Boutron A, Chen YC, Cox JE, Cardon CM, Van Vranken JG, Dephoure N, Redin C, Boudina S, Gygi SP, Brivet M, Thummel CS, Rutter J. A mitochondrial pyruvate carrier required for pyruvate uptake in yeast, Drosophila, and humans. Science. 2012;337:96–100. doi: 10.1126/science.1218099. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- HL007344/NH/NIH HHS/United States
- ES028365/NH/NIH HHS/United States
- R01 DK104998/DK/NIDDK NIH HHS/United States
- AR059190/NH/NIH HHS/United States
- R01 AG060637/AG/NIA NIH HHS/United States
- DK116522/NH/NIH HHS/United States
- DK104998/NH/NIH HHS/United States
- R01 HD084645/HD/NICHD NIH HHS/United States
- HL007638/NH/NIH HHS/United States
- DK092412/NH/NIH HHS/United States
- P30 DK054759/DK/NIDDK NIH HHS/United States
- HD082109/NH/NIH HHS/United States
- HD084645/NH/NIH HHS/United States
- R01 AR071762/AR/NIAMS NIH HHS/United States
- GM007337/NH/NIH HHS/United States
- T32 HL007344/HL/NHLBI NIH HHS/United States
- R00 AR059190/AR/NIAMS NIH HHS/United States
- DK112751/NH/NIH HHS/United States
- HL130146/NH/NIH HHS/United States
- DK101183/NH/NIH HHS/United States
- T32 GM007337/GM/NIGMS NIH HHS/United States
- R01 HD082109/HD/NICHD NIH HHS/United States
LinkOut - more resources
Full Text Sources

