Impact of Huntington's Disease on Mental Rotation Performance in Motor Pre-Symptomatic Individuals
- PMID: 31306138
- PMCID: PMC7266007
- DOI: 10.3233/JHD-190348
Impact of Huntington's Disease on Mental Rotation Performance in Motor Pre-Symptomatic Individuals
Abstract
Background: Huntington's disease (HD) is a genetic disorder known for affecting motor control. Despite evidence for the impact of HD on visual cortico-striatal loops, evidence for impaired visual perception in early symptomatic HD patients is limited; much less is known about what happens during the HD prodrome.
Objective: The goals of this study were to evaluate perceptual processing in motor pre-manifest HD gene-carriers (Pre-HDs) during a visual mental rotation task.
Methods: To achieve this goal, 79 participants including 24 Pre-HD participants and 55 healthy matched controls were scanned using functional MRI as they performed a mental rotation task. Another group of 36 subjects including 15 pre-HDs and 21 healthy age/gender matched controls participated in a control behavioral test of judgment of line orientation outside the scanner.
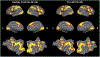
Results: We found that, although Pre-HDs (in this stage of disease) did not demonstrate slower response times, their response accuracy was lower than controls. On the fMRI task, controls showed a significant decrease in activity in the occipito-temporal (OT) visual network and increase in activity in the caudo-fronto-parietal (CFP) network with mental rotation load. Interestingly, the amount of mental rotation-related activity decrease in the OT network was reduced in Pre-HDs compared to controls while, the level of CFP response remained unchanged between the two groups. Comparing the link between the evoked BOLD activity within these networks and response accuracy (i.e., behavior), we found that the models fit to data from controls were less accurate in predicting response accuracy of Pre-HDs.
Conclusion: These findings provide some of the earliest functional evidence of impaired visual processing and altered neural processing underlying visual perceptual decision making during the HD prodrome.
Keywords: Mental rotation; Pre-HD; functional MRI; visual system.
Conflict of interest statement
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Figures
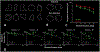

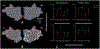




Similar articles
-
Altered brain mechanisms of emotion processing in pre-manifest Huntington's disease.Brain. 2012 Apr;135(Pt 4):1165-79. doi: 10.1093/brain/aws024. Brain. 2012. PMID: 22505631 Free PMC article.
-
Longitudinal changes in the fronto-striatal network are associated with executive dysfunction and behavioral dysregulation in Huntington's disease: 30 months IMAGE-HD data.Cortex. 2017 Jul;92:139-149. doi: 10.1016/j.cortex.2017.04.001. Epub 2017 Apr 15. Cortex. 2017. PMID: 28482181
-
Through your eyes or mine? The neural correlates of mental state recognition in Huntington's disease.Hum Brain Mapp. 2018 Mar;39(3):1354-1366. doi: 10.1002/hbm.23923. Epub 2017 Dec 17. Hum Brain Mapp. 2018. PMID: 29250867 Free PMC article.
-
Aberrant brain network connectivity in presymptomatic and manifest Huntington's disease: A systematic review.Hum Brain Mapp. 2020 Jan;41(1):256-269. doi: 10.1002/hbm.24790. Epub 2019 Sep 18. Hum Brain Mapp. 2020. PMID: 31532053 Free PMC article.
-
Imaging the spin: Disentangling the core processes underlying mental rotation by network mapping of data from meta-analysis.Neurosci Biobehav Rev. 2023 Jul;150:105187. doi: 10.1016/j.neubiorev.2023.105187. Epub 2023 Apr 20. Neurosci Biobehav Rev. 2023. PMID: 37086933 Review.
Cited by
-
The effect of impulsivity and inhibitory control deficits in the saccadic behavior of premanifest Huntington's disease individuals.Orphanet J Rare Dis. 2019 Nov 8;14(1):246. doi: 10.1186/s13023-019-1218-y. Orphanet J Rare Dis. 2019. PMID: 31703597 Free PMC article.
References
-
- Andrew SE, Goldberg YP, Kremer B, Telenius H, Theilmann J, Adam S, et al. The relationship between trinucleotide (CAG) repeat length. Nat Genet. 1993;4:398–403. - PubMed
-
- O’Donovan MC. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell. 1993;72(6):971–83. - PubMed
-
- Kremer B, Goldberg P, Andrew SE, Theilmann J, Telenius H, Zeisler J, et al. A worldwide study of the Huntington’s disease mutation: The sensitivity and specificity of measuring CAG repeats. N Engl J Med. 1994;330(20):1401–6. - PubMed
-
- Huntington G On chorea. The Medical and Surgical Reporter: A Weekly Journal. 1872;26(15):317–21.
-
- Wolf RC, Sambataro F, Vasic N, Baldas EM, Ratheiser I, Bernhard Landwehrmeyer G, et al. Visual system integrity and cognition in early Huntington’s disease. Eur J Neurosci. 2014;40(2):2417–26. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

