A pharmacokinetic and pharmacogenetic evaluation of contraceptive implants and antiretroviral therapy among women in Kenya and Uganda
- PMID: 31306173
- PMCID: PMC6774811
- DOI: 10.1097/QAD.0000000000002308
A pharmacokinetic and pharmacogenetic evaluation of contraceptive implants and antiretroviral therapy among women in Kenya and Uganda
Abstract
Objectives: To evaluate pharmacokinetics and pharmacogenetics of contraceptive implant progestin concentrations in HIV-positive women initiating efavirenz (EFV)-containing or nevirapine (NVP)-containing antiretroviral therapy (ART).
Design: We analyzed stored samples from women self-reporting implant use in the Partners PrEP Study.
Methods: Plasma samples collected every 6 months were analyzed for levonorgestrel and etonogestrel concentrations. Progestin concentrations from samples collected after ART initiation were compared with pre-ART concentrations for intraindividual comparisons. We used adjusted linear mixed models to compare hormone concentrations between individuals on EFV and NVP to a no ART group. We then evaluated whether possessing certain alleles with known or possible influences on EFV, NVP, or progestin metabolism were associated with changes in progestin concentrations or modified the association between ART use and progestin concentrations.
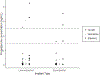
Results: Our analysis included 11 women who initiated EFV, 13 who initiated NVP, and 36 who remained ART-naive. In the EFV group, the adjusted geometric mean ratio (aGMR) of levonorgestrel was 0.39 [90% confidence intervals (0.31, 0.49); P < 0.001] and the etonogestrel aGMR was 0.51 (0.34, 0.76; P = 0.006) compared with the control group. No difference was observed in the NVP group compared with controls [levonorgestrel 0.93 (0.74, 1.18); P = 0.64; etonogestrel 1.07 (0.77, 1.50); P = 0.73]. Possession of four allele variants were found to result in further reductions in progestin concentrations among those receiving EFV.
Conclusion: Concomitant use of EFV significantly reduces levonorgestrel or etonogestrel concentrations by 61 and 49%, respectively, compared with no ART use. We also report allelic variants in hepatic enzymes that influenced the extent of the observed drug-interaction between progestins and EFV.
Figures
Similar articles
-
Efavirenz decreases etonogestrel exposure: a pharmacokinetic evaluation of implantable contraception with antiretroviral therapy.AIDS. 2017 Sep 10;31(14):1965-1972. doi: 10.1097/QAD.0000000000001591. AIDS. 2017. PMID: 28692531 Free PMC article.
-
Unintended Pregnancies Observed With Combined Use of the Levonorgestrel Contraceptive Implant and Efavirenz-based Antiretroviral Therapy: A Three-Arm Pharmacokinetic Evaluation Over 48 Weeks.Clin Infect Dis. 2016 Mar 15;62(6):675-682. doi: 10.1093/cid/civ1001. Epub 2015 Dec 8. Clin Infect Dis. 2016. PMID: 26646680 Free PMC article. Clinical Trial.
-
The Effect of Gene Variants on Levonorgestrel Pharmacokinetics When Combined With Antiretroviral Therapy Containing Efavirenz or Nevirapine.Clin Pharmacol Ther. 2017 Sep;102(3):529-536. doi: 10.1002/cpt.667. Epub 2017 May 30. Clin Pharmacol Ther. 2017. PMID: 28187506 Free PMC article. Clinical Trial.
-
Pregnancy rates in HIV-positive women using contraceptives and efavirenz-based or nevirapine-based antiretroviral therapy in Kenya: a retrospective cohort study.Lancet HIV. 2015 Nov;2(11):e474-82. doi: 10.1016/S2352-3018(15)00184-8. Epub 2015 Oct 22. Lancet HIV. 2015. PMID: 26520927 Free PMC article. Review.
-
Concomitant contraceptive implant and efavirenz use in women living with HIV: perspectives on current evidence and policy implications for family planning and HIV treatment guidelines.J Int AIDS Soc. 2017 May 11;20(1):21396. doi: 10.7448/IAS.20.1.21396. J Int AIDS Soc. 2017. PMID: 28530033 Free PMC article. Review.
Cited by
-
Contraceptive implant use duration is not associated with breakthrough pregnancy among women living with HIV and using efavirenz: a retrospective, longitudinal analysis.J Int AIDS Soc. 2022 Sep;25(9):e26001. doi: 10.1002/jia2.26001. J Int AIDS Soc. 2022. PMID: 36073977 Free PMC article.
-
Effect of Topiramate on Serum Etonogestrel Concentrations Among Contraceptive Implant Users.Obstet Gynecol. 2022 Apr 1;139(4):579-587. doi: 10.1097/AOG.0000000000004697. Epub 2022 Mar 10. Obstet Gynecol. 2022. PMID: 35594123 Free PMC article. Clinical Trial.
-
Effect of double-dose levonorgestrel subdermal implant in women taking efavirenz-based antiretroviral therapy: The DoubLNG pharmacokinetic study.Contraception. 2023 Jun;122:109975. doi: 10.1016/j.contraception.2023.109975. Epub 2023 Feb 12. Contraception. 2023. PMID: 36787829 Free PMC article. Clinical Trial.
-
Comparison of etonogestrel bioanalytical assay results in plasma and serum within and across laboratories.Contraception. 2025 Feb;142:110720. doi: 10.1016/j.contraception.2024.110720. Epub 2024 Oct 2. Contraception. 2025. PMID: 39362338
-
Clinical Relevance of Drug Interactions in People Living with Human Immunodeficiency Virus on Antiretroviral Therapy-Update 2022: Systematic Review.Pharmaceutics. 2023 Oct 18;15(10):2488. doi: 10.3390/pharmaceutics15102488. Pharmaceutics. 2023. PMID: 37896248 Free PMC article. Review.
References
-
- UNAIDS. Global report: UNAIDS report on the global AIDS epidemic 2013 In. Geneva, Switzerland: UNAIDS; 2013.
-
- Desgrees-du-Lou A, Msellati P, Viho I, Yao A, Yapi D, Kassi P, et al. Contraceptive use, protected sexual intercourse and incidence of pregnancies among African HIV-infected women. DITRAME ANRS 049 Project, Abidjan 1995–2000. International Journal of STD & AIDS 2002; 13(7):462–468. - PubMed
-
- Halperin DT, Stover J, Reynolds HW. Benefits and costs of expanding access to family planning programs to women living with HIV. AIDS 2009; 23(Supp 1):S123–S130. - PubMed
-
- UNAIDS. Global plan towards the elimination of new HIV infections In. Geneva, Switzerland; 2011.