A junctophilin-caveolin interaction enables efficient coupling between ryanodine receptors and BKCa channels in the Ca2+ microdomain of vascular smooth muscle
- PMID: 31308177
- PMCID: PMC6721949
- DOI: 10.1074/jbc.RA119.008342
A junctophilin-caveolin interaction enables efficient coupling between ryanodine receptors and BKCa channels in the Ca2+ microdomain of vascular smooth muscle
Abstract
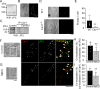
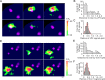
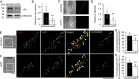
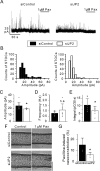
Functional coupling between large-conductance Ca2+-activated K+ (BKCa) channels in the plasma membrane (PM) and ryanodine receptors (RyRs) in the sarcoplasmic reticulum (SR) is an essential mechanism for regulating mechanical force in most smooth muscle (SM) tissues. Spontaneous Ca2+ release through RyRs (Ca2+ sparks) and subsequent BKCa channel activation occur within the PM-SR junctional sites. We report here that a molecular interaction of caveolin-1 (Cav1), a caveola-forming protein, with junctophilin-2 (JP2), a bridging protein between PM and SR, positions BKCa channels near RyRs in SM cells (SMCs) and thereby contributes to the formation of a molecular complex essential for Ca2+ microdomain function. Approximately half of all Ca2+ sparks occurred within a close distance (<400 nm) from fluorescently labeled JP2 or Cav1 particles, when they were moderately expressed in primary SMCs from mouse mesenteric artery. The removal of caveolae by genetic Cav1 ablation or methyl-β-cyclodextrin treatments significantly reduced coupling efficiency between Ca2+ sparks and BKCa channel activity in SMCs, an effect also observed after JP2 knockdown in SMCs. A 20-amino acid-long region in JP2 appeared to be essential for the observed JP2-Cav1 interaction, and we also observed an interaction between JP2 and the BKCa channel. It can be concluded that the JP2-Cav1 interaction provides a structural and functional basis for the Ca2+ microdomain at PM-SR junctions and mediates cross-talk between RyRs and BKCa channels, converts local Ca2+ sparks into membrane hyperpolarization, and contributes to stabilizing resting tone in SMCs.
Keywords: blood pressure; calcium channel; calcium imaging; caveolin; fluorescence resonance energy transfer (FRET); molecular imaging; patch clamp; potassium channel; single-molecule biophysics; vascular smooth muscle cells.
© 2019 Saeki et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures



Similar articles
-
Caveolin-1 facilitates the direct coupling between large conductance Ca2+-activated K+ (BKCa) and Cav1.2 Ca2+ channels and their clustering to regulate membrane excitability in vascular myocytes.J Biol Chem. 2013 Dec 20;288(51):36750-61. doi: 10.1074/jbc.M113.511485. Epub 2013 Nov 7. J Biol Chem. 2013. PMID: 24202214 Free PMC article.
-
Exercise Prevents Upregulation of RyRs-BKCa Coupling in Cerebral Arterial Smooth Muscle Cells From Spontaneously Hypertensive Rats.Arterioscler Thromb Vasc Biol. 2016 Aug;36(8):1607-17. doi: 10.1161/ATVBAHA.116.307745. Epub 2016 Jun 23. Arterioscler Thromb Vasc Biol. 2016. PMID: 27339460
-
Aldosterone-Induced Sarco/Endoplasmic Reticulum Ca2+ Pump Upregulation Counterbalances Cav1.2-Mediated Ca2+ Influx in Mesenteric Arteries.Front Physiol. 2022 Mar 11;13:834220. doi: 10.3389/fphys.2022.834220. eCollection 2022. Front Physiol. 2022. PMID: 35360237 Free PMC article.
-
Elementary calcium signaling in arterial smooth muscle.Channels (Austin). 2019 Dec;13(1):505-519. doi: 10.1080/19336950.2019.1688910. Channels (Austin). 2019. PMID: 31797713 Free PMC article. Review.
-
Role of ryanodine receptor subtypes in initiation and formation of calcium sparks in arterial smooth muscle: comparison with striated muscle.J Biomed Biotechnol. 2009;2009:135249. doi: 10.1155/2009/135249. Epub 2009 Dec 8. J Biomed Biotechnol. 2009. PMID: 20029633 Free PMC article. Review.
Cited by
-
A molecular complex of Cav1.2/CaMKK2/CaMK1a in caveolae is responsible for vascular remodeling via excitation-transcription coupling.Proc Natl Acad Sci U S A. 2022 Apr 19;119(16):e2117435119. doi: 10.1073/pnas.2117435119. Epub 2022 Apr 11. Proc Natl Acad Sci U S A. 2022. PMID: 35412911 Free PMC article.
-
Plasma membrane curvature regulates the formation of contacts with the endoplasmic reticulum.Nat Cell Biol. 2024 Nov;26(11):1878-1891. doi: 10.1038/s41556-024-01511-x. Epub 2024 Sep 17. Nat Cell Biol. 2024. PMID: 39289582 Free PMC article.
-
Coronary Large Conductance Ca2+-Activated K+ Channel Dysfunction in Diabetes Mellitus.Front Physiol. 2021 Oct 21;12:750618. doi: 10.3389/fphys.2021.750618. eCollection 2021. Front Physiol. 2021. PMID: 34744789 Free PMC article. Review.
-
Correlative super-resolution analysis of cardiac calcium sparks and their molecular origins in health and disease.Open Biol. 2023 May;13(5):230045. doi: 10.1098/rsob.230045. Epub 2023 May 24. Open Biol. 2023. PMID: 37220792 Free PMC article.
-
Endoplasmic Reticulum-Plasma Membrane Junctions as Sites of Depolarization-Induced Ca2+ Signaling in Excitable Cells.Annu Rev Physiol. 2023 Feb 10;85:217-243. doi: 10.1146/annurev-physiol-032122-104610. Epub 2022 Oct 6. Annu Rev Physiol. 2023. PMID: 36202100 Free PMC article. Review.
References
-
- Takeshima H., Komazaki S., Nishi M., Iino M., and Kangawa K. (2000) Junctophilins: a novel family of junctional membrane complex proteins. Mol. Cell 6, 11–22 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

