Electroacupuncture treatment upregulates α7nAChR and inhibits JAK2/STAT3 in dorsal root ganglion of rat with spared nerve injury
- PMID: 31308727
- PMCID: PMC6613452
- DOI: 10.2147/JPR.S203867
Electroacupuncture treatment upregulates α7nAChR and inhibits JAK2/STAT3 in dorsal root ganglion of rat with spared nerve injury
Abstract
Background: Neuropathic pain with complicated mechanism severely disrupts patient quality of life. The novel approaches and more effective management should be further investigated. It was reported that alpha-7 nicotinic acetylcholine receptor (α7nAChR) and janus kinase 2 (JAK2)/signal transducer and activator of transcription 3 (STAT3) signaling in dorsal root ganglion (DRG) contributed to the pathogenesis of neuropathic pain. Our previous study has shown that electroacupuncture (EA) alleviated neuropathic pain via activating α7nAChR in the spinal cord. However, whether the effect of 2 Hz EA on spared nerve injury (SNI)-induced neuropathic pain is mediated through modulation of α7nAChR and JAK2/STAT3 pathway in the DRG remains unclear.
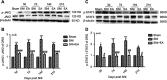
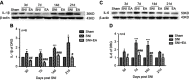
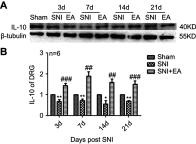
Materials and methods: The SNI-induced neuropathic pain rat model was used in this study. After application of 2 Hz EA treatment to SNI rats on day 3, 7, 14 and 21 post-surgery, the expression levels of α7nAChR, JAK2/STAT3 and some cytokines in DRG were determined by qRT-PCR and Western blot analysis.
Results: We found that SNI induced significant down-regulation of α7nAChR mRNA and protein expression. SNI also obviously elicited the decrease in anti-inflammatory cytokine IL-10 protein expression. The enhancement of p-JAK2, p-STAT3, pro-inflammatory cytokines IL-1β and IL-6 protein levels induced by SNI were also observed. However, 2 Hz EA treatment to SNI rats distinctly improved α7nAChR and IL-10 levels and reduced p-JAK2, p-STAT3, IL-1β and IL-6 expression in the DRG.
Conclusion: Our present study suggested that 2 Hz EA treatment indeed activated α7nAChR, suppressed JAK2/STAT3 signaling and re-balanced the relationship between pro-inflammatory and anti-inflammatory cytokines in DRG of SNI rat, which provided insight into our understanding of the mechanism for 2 Hz EA to attenuate neuropathic pain.
Keywords: JAK2/STAT3; dorsal root ganglion; electroacupuncture; neuropathic pain; α7nAChR.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures

Similar articles
-
Electroacupuncture Modulates Spinal BDNF/TrκB Signaling Pathway and Ameliorates the Sensitization of Dorsal Horn WDR Neurons in Spared Nerve Injury Rats.Int J Mol Sci. 2020 Sep 7;21(18):6524. doi: 10.3390/ijms21186524. Int J Mol Sci. 2020. PMID: 32906633 Free PMC article.
-
Electroacupuncture ameliorates mechanical hypersensitivity by down-regulating spinal Janus kinase 2/signal transducer and activation of transcription 3 and interleukin 6 in rats with spared nerve injury.Acupunct Med. 2021 Aug;39(4):358-366. doi: 10.1177/0964528420938376. Epub 2020 Aug 2. Acupunct Med. 2021. PMID: 32744065
-
Involvement of α7nAChR in electroacupuncture relieving neuropathic pain in the spinal cord of rat with spared nerve injury.Brain Res Bull. 2018 Mar;137:257-264. doi: 10.1016/j.brainresbull.2018.01.002. Epub 2018 Jan 4. Brain Res Bull. 2018. PMID: 29307658
-
[Exploring the mechanism of wrist-ankle acupuncture in treating chemotherapy-induced neuropathic pain in mice by regulating neuroinflammatory responses through α7 nicotinic acetylcholine receptor in the spinal dorsal horn].Zhen Ci Yan Jiu. 2025 Jul 25;50(7):735-742. doi: 10.13702/j.1000-0607.20241267. Zhen Ci Yan Jiu. 2025. PMID: 40691023 Chinese.
-
Electroacupuncture inhibits TLR4/NF-κB signaling in the dorsal root ganglion of rats with spared nerve injury.Acupunct Med. 2024 Oct;42(5):275-284. doi: 10.1177/09645284241279874. Epub 2024 Sep 27. Acupunct Med. 2024. PMID: 39340148
Cited by
-
Electroacupuncture Alleviates Spared Nerve Injury-Induced Neuropathic Pain And Modulates HMGB1/NF-κB Signaling Pathway In The Spinal Cord.J Pain Res. 2019 Oct 16;12:2851-2863. doi: 10.2147/JPR.S220201. eCollection 2019. J Pain Res. 2019. PMID: 31695479 Free PMC article.
-
Reduced vagal tone in women with endometriosis and auricular vagus nerve stimulation as a potential therapeutic approach.Sci Rep. 2021 Jan 14;11(1):1345. doi: 10.1038/s41598-020-79750-9. Sci Rep. 2021. PMID: 33446725 Free PMC article. Clinical Trial.
-
Peripheral Neuropathic Pain: From Experimental Models to Potential Therapeutic Targets in Dorsal Root Ganglion Neurons.Cells. 2020 Dec 21;9(12):2725. doi: 10.3390/cells9122725. Cells. 2020. PMID: 33371371 Free PMC article. Review.
-
Targeting the JAK2/STAT3 signaling pathway for chronic pain.Aging Dis. 2024 Feb 1;15(1):186-200. doi: 10.14336/AD.2023.0515. Aging Dis. 2024. PMID: 37307838 Free PMC article. Review.
-
Electroacupuncture Modulates Spinal BDNF/TrκB Signaling Pathway and Ameliorates the Sensitization of Dorsal Horn WDR Neurons in Spared Nerve Injury Rats.Int J Mol Sci. 2020 Sep 7;21(18):6524. doi: 10.3390/ijms21186524. Int J Mol Sci. 2020. PMID: 32906633 Free PMC article.
References
-
- Burke NN, Geoghegan E, Kerr DM, Moriarty O, Finn DP, Roche M. Altered neuropathic pain behaviour in a rat model of depression is associated with changes in inflammatory gene expression in the amygdala. Genes Brain Behav. 2013;12(7):705–713. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

