Exposure to the smell and taste of milk to accelerate feeding in preterm infants
- PMID: 31311064
- PMCID: PMC6634986
- DOI: 10.1002/14651858.CD013038.pub2
Exposure to the smell and taste of milk to accelerate feeding in preterm infants
Update in
-
Exposure to the smell and taste of milk to accelerate feeding in preterm infants.Cochrane Database Syst Rev. 2024 May 9;5(5):CD013038. doi: 10.1002/14651858.CD013038.pub3. Cochrane Database Syst Rev. 2024. PMID: 38721883 Free PMC article.
Abstract
Background: Preterm infants are often unable to co-ordinate sucking, swallowing and breathing for oral feeding because of their immaturity; in such cases, initial nutrition is provided by orogastric or nasogastric tube feeding. Feed intolerance is common and can delay attainment of full enteral feeds and sucking feeds, which prolongs the need for intravenous nutrition and hospital stay. Smell and taste play an important role in the activation of physiological pre-absorptive processes that contribute to food digestion and absorption. However, during tube feedings, milk bypasses the nasal and oral cavities, which limits exposure to the smell and taste of milk. Provision of the smell and taste of milk with tube feedings is non-invasive and inexpensive; and if it does accelerate the transition to enteral feeds, and then to sucking feeds, it would be of considerable potential benefit to infants, their families, and the healthcare system.
Objectives: To assess whether exposure to the smell or taste (or both) of milk administered with tube feedings can accelerate progress to full sucking feeds without adverse effects in preterm infants.
Search methods: We used the standard search strategy of Cochrane Neonatal to search the Cochrane Central Register of Controlled Trials (CENTRAL 2018, Issue 5), MEDLINE via PubMed (1966 to 1 June 2018), Embase (1980 to 1 June 2018), and CINAHL (1982 to 1 June 2018). We also searched clinical trials databases, conference proceedings, and the reference lists of retrieved articles for randomised and quasi-randomised trials.
Selection criteria: We included randomised and quasi-randomised studies that compared the provision of the smell or taste of milk (or both) immediately before or at the time of tube feedings, with no provision of smell or taste.
Data collection and analysis: Two review authors independently abstracted data according to Cochrane Neonatal methodology; they also assessed risk of bias, and the quality of evidence at the outcome level using the GRADE approach. We performed meta-analyses using risk ratio (RR) for dichotomous data and mean difference (MD) for continuous data, with their respective 95% confidence intervals (CIs).
Main results: Three trials involving a total of 161 preterm infants were included in this review, but only two trials (131 infants) contributed data for meta-analysis. There was no evidence of a clear effect of exposure to the smell and taste of milk with tube feedings on time taken to reach full sucking feeds (MD -2.57 days, 95% CI -5.15 to 0.02; I2 = 17%; 2 trials, 131 infants; very low-quality evidence). One trial reported no adverse effects. There was no evidence of a clear effect of exposure to the smell and taste of milk on the following outcomes: time taken to reach full enteral feeds (MD -1.57 days, 95% CI -6.25 to 3.11; 1 trial, 51 infants; very low-quality evidence), duration of parenteral nutrition (MD -2.20 days, 95% CI -9.49 to 5.09; 1 trial, 51 infants; very low-quality evidence), incidence of necrotising enterocolitis (RR 0.62, 95% CI 0.15 to 2.48; 1 trial, 51 infants; low-quality evidence), and late infection (RR 2.46, 95% CI 0.27 to 22.13; 1 trial, 51 infants; low-quality evidence). There was very low-quality evidence demonstrating that exposure to the smell and taste of milk decreased duration of hospitalisation by almost four days (MD -3.89 days, 95% CI -7.03 to -0.75; I2 = 51%; 2 trials, 131 infants). In two trials, an increased growth velocity was noted in infants exposed to the intervention, but we were unable to combine data to perform meta-analysis. No data were available to assess feed intolerance and rates of exclusive breastfeeding at discharge. Included trials were small and had methodological limitations including lack of randomisation (one trial), lack of blinding, and different inclusion criteria and administration of the interventions.
Authors' conclusions: Evidence from two trials suggests that exposure to the smell and taste of milk with tube feedings has no clear effect on time taken to reach full sucking feeds, but it may decrease length of hospitalisation. However, these results are uncertain due to the very low quality of the evidence. There is also limited evidence about the impact on other important clinical outcomes and on safety. Future research should examine the effect of exposure to the smell and taste of milk with tube feedings on clinical outcomes during hospitalisation, such as attainment of full enteral and sucking feeds, safety, feed tolerance, incidence of infection, and infant growth. Additionally, future research should be sufficiently powered to evaluate the effect of the intervention in infants of different gestational ages, on each sex separately, and on the optimal frequency and duration of exposure.
Conflict of interest statement
Frank Bloomfield and Jane Harding have designed, and are steering committee members of, a randomised trial of different nutritional approaches to feeding moderate‐ to late‐preterm infants that includes provision of smell and taste as one intervention. Mariana Muelbert is one of the research team members on this trial (DIAMOND Trial, Australian New Zealand Clinical Trials Registry ACTRN12616001199404). Frank Bloomfield is a co‐author of one of the trials included in this review (Beker 2017a), but had no role in the assessment, data extraction, or analysis of data from this trial for this review.
There is no other conflict, and in particular no benefits of any kind have been received by the authors in relation to any element of the proposed review.
Mariana Muelbert: none known.
Jane Harding: none known.
Frank Bloomfield: none known.
Luling Lin: none known.
Figures
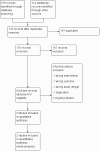








References
References to studies included in this review
Beker 2017a {published data only}
Davidson 2015 {published data only}
-
- Davidson J, Hwang JS, Kosow M, Maron JL. Optimal timing and sex specific responses to olfactory stimulation with mother's own milk to decrease the time to full oral feeds in premature infants. Pediatric Academic Societies (PAS) Annual Meeting. San Diego, California, US, 2015.
Yildiz 2011 {published data only}
-
- Yildiz A, Arikan D, Gozum S, Tastekin A, Budancamanak I. The effect of the odor of breast milk on the time needed for transition from gavage to total oral feeding in preterm infants. Journal of Nursing Scholarship 2011;43(3):265‐73. [DOI: 10.1111/j.1547-5069.2011.01410.x; PUBMED: 21884372] - DOI - PubMed
References to studies excluded from this review
Beker 2016 {published data only}
Bingham 2003 {published data only}
Neshat 2016 {published data only}
-
- Neshat H, Jebreili M, Seyyedrasouli A, Ghojazade M, Hosseini MB, Hamishehkar H. Effects of breast milk and vanilla odors on premature neonate's heart rate and blood oxygen saturation during and after venipuncture. Pediatrics and Neonatology 2016;57(3):225‐31. [DOI: 10.1016/j.pedneo.2015.09.004; PUBMED: 26560183] - DOI - PubMed
References to ongoing studies
ACTRN12617000583347 {published data only}
-
- ACTRN12617000583347. Effect of smell and taste to improve nutrition in very preterm babies [Smell and taste with tube feeding to improve nutrition in very preterm infants: a randomized controlled trial]. www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=372308 (first received 19 April 2017).
Bloomfield 2018 {published data only}
-
- Bloomfield FH, Harding JE, Meyer MP, Alsweiler JM, Jiang Y, Wall CR, et al. The DIAMOND trial – DIfferent Approaches to MOderate & late preterm Nutrition: Determinants of feed tolerance, body composition and development: protocol of a randomised trial. BMC Pediatrics 2018;18(1):220. [DOI: 10.1186/s12887-018-1195-7] - DOI - PMC - PubMed
Additional references
ANZNN 2016
-
- Australian and New Zealand Neonatal Network. ANZNN 2017 Data Dictionary. anznn.net/Portals/0/DataDictionaries/ANZNN_2017_Data_Dictionary.pdf (accessed May 2018).
Bloomfield 2017
Cormack 2016
Covidence 2018 [Computer program]
-
- Veritas Health Innovation. Covidence. Version accessed 1 July 2018. Melbourne, Australia: Veritas Health Innovation.
Dasgupta 2016
Deeks 2017
-
- Deeks JJ, Higgins JP, Altman DG editor(s). Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JP, Churchill R, Chandler J, Cumpston MS editor(s). Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017). The Cochrane Collaboration, 2017. Available from www.training.cochrane.org/handbook.
Fanaro 2013
Gargasz 2012
GRADEpro GDT [Computer program]
-
- McMaster University (developed by Evidence Prime). GRADEpro GDT. Version accessed 19 January 2018. Hamilton (ON): McMaster University (developed by Evidence Prime), 2015.
Higgins 2011
-
- Higgins JP, Deeks JJ, Altman DG, editor(s). Chapter 16: Special topics in statistics. In: Higgins JP, Green S editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Higgins 2017
-
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Churchill R, Chandler J, Cumpston MS (editors), Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017). The Cochrane Collaboration, 2017. Available from www.training.cochrane.org/handbook.
Johnson 2014
-
- Johnson J, Patel AL, Bigger HR, Engstrom JL, Meier PP. Economic benefits and costs of human milk feedings: a strategy to reduce the risk of prematurity‐related morbidities in very‐low‐birth‐weight infants. Advances in Nutrition 2014;5(2):207‐12. [DOI: 10.3945/an.113.004788; PUBMED: 24618763] - DOI - PMC - PubMed
Lipchock 2011
Marlier 1998
Mattes 1997
Moher 2009
Moore 2011
Patel 2005
Power 2008
Review Manager 2014 [Computer program]
-
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Sarnat 1978
Schünemann 2013
-
- Schünemann H, Brożek J, Guyatt G, Oxman A, editor(s). Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach (updated October 2013). GRADE Working Group, 2013. gdt.guidelinedevelopment.org/app/handbook/handbook.html (accessed 18 May 2018).
Smeets 2010
The SIFT Investigators Group 2013
Toce 1987
Walsh 1986
WHO 2008
-
- World Health Organization. Indicators for assessing infant and young child feeding practices ‐ part I: definition. www.who.int/maternal_child_adolescent/documents/9789241596664/en/ (assessed 18 May 2018):1‐19.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

