Successful Reconstruction of the Right Ventricular Outflow Tract by Implantation of Thymus Stem Cell Engineered Graft in Growing Swine
- PMID: 31312760
- PMCID: PMC6609916
- DOI: 10.1016/j.jacbts.2019.02.001
Successful Reconstruction of the Right Ventricular Outflow Tract by Implantation of Thymus Stem Cell Engineered Graft in Growing Swine
Abstract
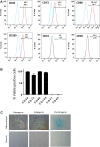
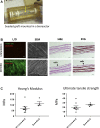
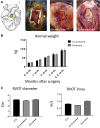
Graft cellularization holds great promise in overcoming the limitations associated with prosthetic materials currently used in corrective cardiac surgery. In this study, the authors evaluated the advantages of graft cellularization for right ventricular outflow tract reconstruction in a novel porcine model. After 4.5 months from implantation, improved myocardial strain, better endothelialization and cardiomyocyte incorporation, and reduced fibrosis were observed in the cellularized grafts compared with the acellular grafts. To the authors' knowledge, this is the first demonstration of successful right ventricular outflow tract correction using bioengineered grafts in a large animal model.
Keywords: CM, cardiomyocyte; Cx-43, connexin-43; DMEM, Dulbecco’s modified Eagle’s medium; EC, endothelial cell; FBS, fetal bovine serum; IL, interleukin; IsoB4, isolectin B4; MSC, mesenchymal stem cell; PBS, phosphate-buffered saline; PS, penicillin/streptomycin; RT, room temperature; RV, right ventricular; RVOT, right ventricular outflow tract; RVOT-MS, fractional area of change in the right ventricular outflow tract; SIS-ECM, small intestinal submucosa–derived extracellular matrix; T-MSC, thymus-derived mesenchymal stem cell; VMSC, vascular smooth muscle cell; cMYH, cardiac myosin heavy chain; congenital heart disease; reconstruction; right ventricular outflow swine model; tissue engineering; tract stem cells.
Figures











References
-
- Scholl F.G., Boucek M.M., Chan K.C., Valdes-Cruz L., Perryman R. Preliminary experience with cardiac reconstruction using decellularized porcine extracellular matrix scaffold: human applications in congenital heart disease. World J Pediatr Congenit Heart Surg. 2010;1:132–136. - PubMed
-
- Iacobazzi D., Swim M.M., Albertario A., Caputo M., Ghorbel M.T. Thymus-derived mesenchymal stem cells for tissue engineering clinical-grade cardiovascular grafts. Tissue Eng Part A. 2018;24:794–808. - PubMed
-
- Dohmen P.M., da Costa F., Yoshi S. Histological evaluation of tissue-engineered heart valves implanted in the juvenile sheep model: is there a need for in-vitro seeding? J Heart Valve Dis. 2006;15:823–829. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

