A Nitric Oxide-Dependent Presynaptic LTP at Glutamatergic Synapses of the PVN Magnocellular Neurosecretory Cells in vitro in Rats
- PMID: 31316353
- PMCID: PMC6610542
- DOI: 10.3389/fncel.2019.00283
A Nitric Oxide-Dependent Presynaptic LTP at Glutamatergic Synapses of the PVN Magnocellular Neurosecretory Cells in vitro in Rats
Abstract
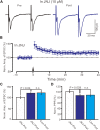
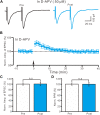
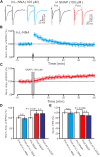
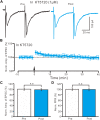
The magnocellular neurosecretory cells (MNCs) of the hypothalamic paraventricular nucleus (PVN) integrate incoming signals to secrete oxytocin (OT), and vasopressin (VP) from their nerve terminals in the posterior pituitary gland. In the absence of gamma-aminobutyric acid A (GABAA) and cannabinoids 1 (CB1) receptor activity, we used whole-cell patch-clamp recording, single-cell reverse transcription-multiplex polymerase chain reaction (SC-RT-mPCR), biocytin histochemistry and pharmacological methods to examine the mechanism of high frequency stimulus (HFS, 100 Hz)-induced long-term potentiation (LTP) at glutamatergic synapses in the PVN MNCs of juvenile male rats. Our results showed that HFS-induced LTP at glutamatergic synapses was accompanied by a decrease in the paired-pulse ratio (PPR) of the PVN MNCs. In these MNCs, HFS-induced LTP persisted in the presence of a group 1 metabotropic glutamate receptor (mGluR1) antagonist; however, it was abolished by an N-methyl-D-aspartic acid (NMDA) receptor blocker. Notably, HFS-induced LTP in the PVN MNCs was completely prevented by a nitric oxide synthase (NOS) inhibitor. The application of an NO donor not only induced the LTP of excitatory glutamatergic inputs in the PVN MNCs, but also occluded the HFS-induced LTP in these MNCs. Moreover, HFS-induced LTP in the PVN MNCs was also abolished by a specific protein kinase A (PKA) inhibitor, KT5720. SC-RT-mPCR analysis revealed that 64.5% (62/96) of MNCs expressed OT mRNA. Our results indicate that a HFS can induce an NMDA receptor and NO cascades dependent on presynaptic glutamatergic LTP in the PVN MNCs via a PKA signaling pathway.
Keywords: NMDA receptor; hypothalamic paraventricular nucleus; long-term synaptic plasticity; nitric oxide; protein kinase A; whole-cell recording.
Figures



References
-
- Bhat G. K., Mahesh V. B., Lamar C. A., Ping L., Aguan K., Brann D. W. (1995). Histochemical ocalization of nitric oxide neurons in the hypothalamus: association with gonadotropin-releasing hormone neurons and colocalization with N-methyl-D-aspartate receptors. Neuroendocrinology 62 187–197. 10.1159/000127004 - DOI - PubMed
LinkOut - more resources
Full Text Sources

