Sensitivity of indocyanine green angiography compared to fluorescein angiography and enhanced depth imaging optical coherence tomography during tapering and fine-tuning of therapy in primary stromal choroiditis: A case series
- PMID: 31317097
- PMCID: PMC6611918
- DOI: 10.1016/j.joco.2018.12.006
Sensitivity of indocyanine green angiography compared to fluorescein angiography and enhanced depth imaging optical coherence tomography during tapering and fine-tuning of therapy in primary stromal choroiditis: A case series
Abstract
Purpose: To investigate indocyanine green angiography (ICGA), fluorescein angiography (FA), and enhanced depth imaging optical coherence tomography measured choroidal thickness (EDI-OCT-CT) in the follow-up of inflammatory activity in stromal choroiditis [Vogt-Koyanagi-Harada disease (VKH) and birdshot retinochoroiditis (BRC)] under treatment in order to monitor tapering of therapy or readjustment of therapy in case of subclinical disease recurrence.
Methods: Patients with initial onset disease and/or treatment-naive stromal choroiditis (VKH & BRC) at entry, quiet under therapy, and having had a follow-up of at least four years monitored with dual FA and ICGA and EDI-OCT-CT measurements were analyzed retrospectively. ICGA and FA scores and EDI-OCT-CT values were correlated with therapy, and significant changes of each modality were correlated with disease evolution.
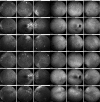
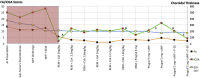
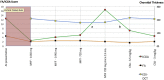
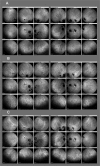
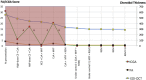
Results: Of the 31 VKH and 29 BRC patients seen from 1995 to 2017 in our center, four patients (2 VKH and 2 BRC patients) fulfilled the inclusion criteria. During tapering, two patients (both VKH) showed no significant ICGA, FA, and EDI-OCT-CT changes (mean follow-up time 5.6 years) and allowed for safe tapering. In the other two (BRC) patients (mean follow-up time 6.25 years), a total of seven significant subclinical changes were demonstrated by ICGA alone after therapy modifications due to side-effects or during attempted tapering of therapy, while FA and EDI-OCT-CT remained unchanged.
Conclusions: ICGA was the most sensitive monitoring modality of stromal choroiditis, able to identify subclinical recurrences following change of therapy and inversely treatment responses after readjusted therapy, events otherwise missed by FA and EDI-OCT. ICGA proved efficient for safe therapy tapering or for timely adjustment of therapy in stromal choroiditis when necessary.
Keywords: Birdshot retinochoroiditis; Indocyanine green angiography; Stromal choroiditis; Treatment; VKH; Vogt-Koyanagi-Harada disease.
Figures

References
-
- Bouchenaki N.H.C. Stromal choroiditis. In: Pleyer U., Mondino B., editors. Essentials in Ophthalmology: Uveitis and Immunological Disorders. Springer; Berlin Heidelberg, New York: 2004. pp. 234–253.
-
- Balci O., Gasc A., Jeannin B., Herbort C.P., Jr. Enhanced depth imaging is less suited than indocyanine green angiography for close monitoring of primary stromal choroiditis: a pilot report. Int Ophthalmol. 2017;37(3):737–748. - PubMed
-
- Balci O., Jeannin B., Herbort C.P., Jr. Contribution of dual fluorescein and indocyanine green angiography to the appraisal of posterior involvement in birdshot retinochoroiditis and Vogt-Koyanagi-Harada disease. Int Ophthalmol. 2018;38(2):527–539. - PubMed
-
- Reddy A.K., Gonzalez M.A., Henry C.R., Yeh S., Sobrin L., Albini T.A. Diagnostic sensitivity of indocyanine green angiography for birdshot chorioretinopathy. JAMA Ophthalmol. 2015;133(7):840–843. - PubMed
-
- Yannuzzi L.A. Indocyanine green angiography: a perspective on use in the clinical setting. Am J Ophthalmol. 2011;151(5):745–751. e741. - PubMed
LinkOut - more resources
Full Text Sources

