Weiqi Decoction Attenuated Chronic Atrophic Gastritis with Precancerous Lesion through Regulating Microcirculation Disturbance and HIF-1 α Signaling Pathway
- PMID: 31320912
- PMCID: PMC6610735
- DOI: 10.1155/2019/2651037
Weiqi Decoction Attenuated Chronic Atrophic Gastritis with Precancerous Lesion through Regulating Microcirculation Disturbance and HIF-1 α Signaling Pathway
Abstract
Aim: Chronic atrophic gastritis (CAG), the precancerous lesions of gastric cancer, plays an important role in the stepwise process of gastric cancer. The ancient Chinese medicine believes in that Qi deficiency and blood stasis are involved in the pathogenesis of CAG. Weiqi decoction, a classical formula from Longhua Hospital, could supplement Qi and activate blood circulation of human beings and has been used for treating CAG in clinic over twenty years. The study aims to clarify the effect and underlying molecular mechanism of Weiqi decoction on CAG rats.
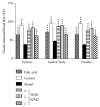
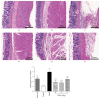
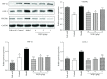
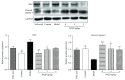
Methods: Forty-eight male Wistar rats were divided randomly into six groups: control group, model group, folic acid group, and WQD-treated groups at doses of 4 g/kg, 2 g/kg, and 1 g/kg, with eight rats in each group. MNNG and saturated NaCl were used to induce CAG rat with precancerous lesion (intestinal metaplasia and dysplasia). After 40 weeks, gastric mucosal blood flow was measured using Laser Doppler Flowmetry. The pathological changes of the gastric mucosa were identified by H&E staining and AB-PAS staining. The protein expression of COX-2, HIF-1α, VEGFR1, VEGFR2, Ki67, and cleaved caspase 3 in the gastric tissues was measured by western blotting approach. Gene expression of COX-2, HIF-1α, VEGF, VEGFR1, VEGFR2, Ang-1, and Ang-2 was detected by using Quantitative PCR method. The PGE2 concentrations in serum were detected by ELISA method. The protein expression of Ki67 in gastric mucosa was also detected by immunohistochemistry.
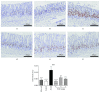
Results: Compared with control rats, atrophy and intestinal metaplasia as well as the microcirculation disturbance of gastric mucosa were induced in the stomach of CAG rats identified by the H&E and AB-PAS staining as well as microcirculation measurement, which could be significantly attenuated by WQD treatment. Moreover, compared with the control group, the protein and gene expression of COX-2, HIF-1α, VEGFR1, and VEGFR2 in gastric tissues of pylorus was obviously increased and the serum PGE2 level was significantly deceased in CAG rats, which could be significantly counteracted by WQD administration. However, the gene expression of Ang-1 and Ang-2 was not significant difference between control rats and CAG rats, and WQD also had no significant effect on the gene expression of Ang-1 and Ang-2. Furthermore, the increased cell proliferation marked by upregulated protein expression of Ki67 and decreased cell apoptosis marked by downregulated protein expression of cleaved caspase 3 in stomach of pylorus in CAG rats were obviously reversed by WQD treatment.
Conclusion: WQD attenuated CAG with precancerous lesion through regulating gastric mucosal blood flow disturbance and HIF-1α signaling pathway.
Figures



Similar articles
-
Chronic atrophic gastritis and intestinal metaplasia induced by high-salt and N-methyl-N'-nitro-N-nitrosoguanidine intake in rats.Exp Ther Med. 2021 Apr;21(4):315. doi: 10.3892/etm.2021.9746. Epub 2021 Feb 3. Exp Ther Med. 2021. PMID: 33717258 Free PMC article.
-
Integrative pharmacological analysis of modified Zuojin formula: Inhibiting the HIF-1α-mediated glycolytic pathway in chronic atrophic gastritis.J Ethnopharmacol. 2025 Jan 13;339:119136. doi: 10.1016/j.jep.2024.119136. Epub 2024 Nov 29. J Ethnopharmacol. 2025. PMID: 39577677
-
Jianwei Xiaoyan granule ameliorates chronic atrophic gastritis by regulating HIF-1α-VEGF pathway.J Ethnopharmacol. 2024 Nov 15;334:118591. doi: 10.1016/j.jep.2024.118591. Epub 2024 Jul 16. J Ethnopharmacol. 2024. PMID: 39025161
-
Gastric atrophy, metaplasia, and dysplasia: a clinical perspective.J Clin Gastroenterol. 2003 May-Jun;36(5 Suppl):S29-36; discussion S61-2. doi: 10.1097/00004836-200305001-00006. J Clin Gastroenterol. 2003. PMID: 12702963 Review.
-
Effects of oxidative stress regulation in inflammation-associated gastric cancer progression treated using traditional Chinese medicines: A review.Medicine (Baltimore). 2023 Nov 17;102(46):e36157. doi: 10.1097/MD.0000000000036157. Medicine (Baltimore). 2023. PMID: 37986311 Free PMC article. Review.
Cited by
-
The effect of phytochemicals in N-methyl-N-nitro-N-nitroguanidine promoting the occurrence and development of gastric cancer.Front Pharmacol. 2023 Jun 29;14:1203265. doi: 10.3389/fphar.2023.1203265. eCollection 2023. Front Pharmacol. 2023. PMID: 37456745 Free PMC article. Review.
-
Global trend and future landscape of intestinal microcirculation research from 2000 to 2021: A scientometric study.World J Gastroenterol. 2023 Mar 7;29(9):1523-1535. doi: 10.3748/wjg.v29.i9.1523. World J Gastroenterol. 2023. PMID: 36998427 Free PMC article.
-
Traditional Chinese medicine for chronic atrophic gastritis: Efficacy, mechanisms and targets.World J Gastroenterol. 2025 Mar 7;31(9):102053. doi: 10.3748/wjg.v31.i9.102053. World J Gastroenterol. 2025. PMID: 40061592 Free PMC article. Review.
-
Amalgamated Pharmacoinformatics Study to Investigate the Mechanism of Xiao Jianzhong Tang against Chronic Atrophic Gastritis.Curr Comput Aided Drug Des. 2024;20(5):598-615. doi: 10.2174/1573409919666230720141115. Curr Comput Aided Drug Des. 2024. PMID: 37475552
-
Jianpiyiqi formula ameliorates chronic atrophic gastritis in rats by modulating the Wnt/β-catenin signaling pathway.Exp Ther Med. 2021 Aug;22(2):878. doi: 10.3892/etm.2021.10310. Epub 2021 Jun 15. Exp Ther Med. 2021. PMID: 34194556 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

