Synthesis and characterization of tetra-ganciclovir cobalt (II) phthalocyanine for electroanalytical applications of AA/DA/UA
- PMID: 31321325
- PMCID: PMC6612534
- DOI: 10.1016/j.heliyon.2019.e01946
Synthesis and characterization of tetra-ganciclovir cobalt (II) phthalocyanine for electroanalytical applications of AA/DA/UA
Abstract
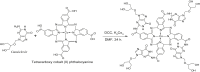
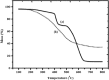
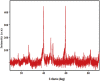
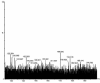
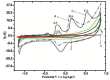
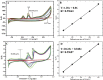
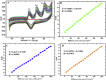
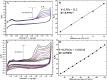
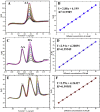
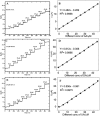
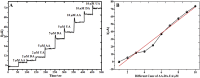
Cobalt (II) phthalocyanine embedded with ganciclovir units has been synthesized by a novel method using tetracarboxylic phthalocyanine reported for the first time. The synthesized dark green colored complexes were characterized by electronic spectroscopy, elemental analysis, FT-IR, MASS and XRD. Thermal stability study reveals that the newly synthesized complex was stable up to 300 °C and XRD patterns showed amorphous nature of the complex. In the present work, the synthesized complex was characterized by cyclic voltammetry and shows the redox behavior corresponding to central metal (Co+II/Co+I) of the complex. Three biomolecules are well-separated by their oxidation peaks in simultaneous determination predicting the potentials for (-128, 335, and 723 mV) with highly increasing current. The low detection limit of AA, DA, and UA were 0.33, 0.03 and 0.10 μmol by CV method and good responses of amperometric and DPV technique. The modified tetra substituted CoTGPc/GCE exhibit an excellent electrocatalytic activity, stability, high sensitivity, good linearity, and selectivity without losing its catalytic activity and proves to be a versatile chemical sensor for commercial pharmaceutical samples, vitamin C tablets, and dopamine injections.
Keywords: Amperometry sensors; Analytical chemistry; Ascorbic acid; CoTGPc; Dopamine; Electrochemistry; Uric acid.
Figures
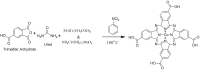





Similar articles
-
Lithium cobalt phosphate electrode for the simultaneous determination of ascorbic acid, dopamine, and serum uric acid by differential pulse voltammetry.Mikrochim Acta. 2021 May 15;188(6):190. doi: 10.1007/s00604-021-04839-5. Mikrochim Acta. 2021. PMID: 33991256
-
ZnO-CuxO/polypyrrole nanocomposite modified electrode for simultaneous determination of ascorbic acid, dopamine, and uric acid.Anal Biochem. 2015 Mar 15;473:53-62. doi: 10.1016/j.ab.2014.12.013. Epub 2015 Jan 7. Anal Biochem. 2015. PMID: 25576954
-
Carbon Black-Carbon Nanotube Co-Doped Polyimide Sensors for Simultaneous Determination of Ascorbic Acid, Uric Acid, and Dopamine.Materials (Basel). 2018 Sep 12;11(9):1691. doi: 10.3390/ma11091691. Materials (Basel). 2018. PMID: 30213063 Free PMC article.
-
Composites of Bimetallic Platinum-Cobalt Alloy Nanoparticles and Reduced Graphene Oxide for Electrochemical Determination of Ascorbic Acid, Dopamine, and Uric Acid.Sci Rep. 2019 Aug 22;9(1):12258. doi: 10.1038/s41598-019-48802-0. Sci Rep. 2019. PMID: 31439896 Free PMC article.
-
One-dimensional NiFe2O4 nanorods modified with sulfur-rich spherical carbon nanoparticles for simultaneous voltammetric determination of ascorbic acid, dopamine and uric acid.Mikrochim Acta. 2019 Jun 13;186(7):434. doi: 10.1007/s00604-019-3496-4. Mikrochim Acta. 2019. PMID: 31197533
Cited by
-
Novel decorated aluminium(iii) phthalocyanine complex with the application of MWCNTs on electrodes: electrochemical non-enzymatic oxidation and reduction of glucose and hydrogen peroxide.RSC Adv. 2023 Jul 11;13(30):20723-20736. doi: 10.1039/d3ra02617e. eCollection 2023 Jul 7. RSC Adv. 2023. PMID: 37441052 Free PMC article.
-
Electrochemical fabrication of Co(OH)2 nanoparticles decorated carbon cloth for non-enzymatic glucose and uric acid detection.Mikrochim Acta. 2022 Sep 20;189(10):385. doi: 10.1007/s00604-022-05437-9. Mikrochim Acta. 2022. PMID: 36125554
-
Electrochemical energy storage in an organic supercapacitor via a non-electrochemical proton charge assembly.Chem Sci. 2023 Dec 19;15(5):1726-1735. doi: 10.1039/d3sc05639b. eCollection 2024 Jan 31. Chem Sci. 2023. PMID: 38303938 Free PMC article.
-
Novel Schiff base cobalt(ii) phthalocyanine with appliance of MWCNTs on GCE: enhanced electrocatalytic activity behaviour of α-amino acids.RSC Adv. 2021 May 6;11(27):16736-16746. doi: 10.1039/d1ra01815a. eCollection 2021 Apr 30. RSC Adv. 2021. PMID: 35479120 Free PMC article.
References
-
- Heien M.L.A.V., Khan A.S., Ariansen J.L., Cheer J.F., Phillips P.E.M., Wight-man R.M. Vol. 102. 2005. Real-time measurement of dopamine fluctuations after cocaine in the brain of behaving rats; pp. 10023–10028. (Proceedings of the National Academy of Sciences of theUnited States of America). - PMC - PubMed
-
- Mounesh, Jilani B.S., Malatesh P., Venugopala Reddy K.R., Lokesh K.S. Simultaneous and sensitive detection of ascorbic acid in presence of dopamine using MWCNTs-decorated cobalt (II) phthalocyanine modified GCE. Microchem. J. 2019;147:755–763.
-
- Arrigoni O., Tullio M.C.D. Ascorbic acid: much more than just an antioxidant. Biochim. Biophys. Acta. 2002;1569:1–9. - PubMed
-
- Kalimuthu P., John S.A. Electropolymerized film of functionalized thiadiazoleon glassy carbon electrode for the simultaneous determination of ascorbic acid, dopamine and uric acid. Bioelectrochem. 2009;77:13–18. - PubMed
-
- Kim J.H., Auerbach J.M., Gómez J.A.R., Velasco I., Gavin D., Lumelsky N., Lee S.H., Nguyen J., Pernaute R.S., Bankiewicz K., McKay R. Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson’s disease. Nature. 2002;418:50–56. - PubMed
LinkOut - more resources
Full Text Sources

