Tisagenlecleucel in relapsed/refractory diffuse large B-cell lymphoma patients without measurable disease at infusion
- PMID: 31332046
- PMCID: PMC6650727
- DOI: 10.1182/bloodadvances.2019000151
Tisagenlecleucel in relapsed/refractory diffuse large B-cell lymphoma patients without measurable disease at infusion
Abstract
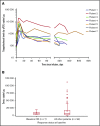
Tisagenlecleucel demonstrated high rates of durable responses in adult patients with relapsed or refractory diffuse large B-cell lymphoma (r/r DLBCL) in the JULIET trial. Most patients (92%) received bridging therapies to control disease after study entry and before tisagenlecleucel infusion. Here, we examine the efficacy and safety of tisagenlecleucel in the subset of 7 patients who achieved complete response (CR) after bridging therapy and before tisagenlecleucel infusion. Tisagenlecleucel rapidly expanded in all 7 patients, and the transgene levels were measurable for up to 2 years after infusion. After infusion, all 7 patients were still in CR at the month 3 evaluation, and 5 of 7 patients remained progression-free >12 months. Adverse events were similar to the overall JULIET population. Cytokine release syndrome (CRS) was reported in 4 of 7 patients (grade 2 = 2 and grade 3 = 2 using the Penn grading scale), and 1 patient experienced grade 1 neurotoxicity. No patient required tocilizumab or steroids for CRS management. These data provide preliminary evidence of tisagenlecleucel efficacy in patients with r/r DLBCL without detectable disease after bridging or salvage therapies and warrant further investigation of tisagenlecleucel as consolidative therapy in future trials. This trial was registered at www.clinicaltrials.gov as #NCT02445248.
© 2019 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: M.R.B. provided consultant services to, was a member of an entity’s board of directors or advisory committees for, and received research funding from Novartis, Kite Pharma, and Juno Therapeutics; provided consultant services to and received honoraria from Novartis, Kite Pharma, Juno Therapeutics, Agios Pharmaceuticals, and CRISPR Therapeutics; was on a speaker’s bureau for, and received travel and honoraria from, Celgene, Kite Pharma, and Agios Pharmaceuticals; and was employed by, provided consultant services to, and received honoraria from Optum. R.T.M. received honoraria from, was a member of an entity’s board of directors or advisory committees for (Scientific Steering Committee for JULIET), and received research funding from Novartis; provided consultant services to and received honoraria from Incyte Inc, Celgene/Juno Therapeutics, and CRISPR Therapeutics; received honoraria from Kite Therapeutics; has patents for and received royalties from Athersys, Inc; was employed by Oregon Health & Science University (OHSU); and provided consultant services to, and received payment from, Novartis (this potential conflict of interest has been reviewed and managed by OHSU). E.K.W. provided consultant services to, was a member of an entity’s board of directors or advisory committees for, and received research funding from Novartis; received travel expenses from European Hematology Association; received research funding from Pharmacyclics and Celldex; provided consultant services to Kalytera; and provided consultant services to and had equity ownership in Cambium Medical Technologies and Cambium Oncology. U.J. provided consultant services to, received honoraria from, was a member of an entity’s board of directors or advisory committees for, and received research funding from Roche and Gilead; provided consultant services to, received honoraria from, and was a member of an entity’s board of directors or advisory committees for Janssen and Celgene; provided consultant services to and received honoraria from AbbVie; was a member of an entity’s board of directors or advisory committees for and received research funding from Novartis; was a member of an entity’s board of directors or advisory committees for Mundipharma, Takeda-Millennium, Amgen, AOP Orphan, GSK, Infinity, and Bioverativ; and research funding from Merck Sharp & Dohme Corporation. J.R.W. was a member of an entity’s board of directors or advisory committees for Novartis, Apotex, Kite Pharma, and Celgene. J.P.M. received honoraria, travel accommodations, and expenses from, and was a speaker for, Kite Pharma; received honoraria from, was a speaker for, and received research funding from Novartis; and received research funding from Fresenius Biotech, Astellas Pharma, Bellicum Pharmaceuticals, Gamida Cell, and Pluristem Ltd. I.F. provided consultant services to AbbVie, Novartis, Merck, Janssen, Seattle Genetics, Gilead, Lundbeck, F. Hoffmann–La Roche Ltd, and Celgene. H.H. was a member of an entity’s board of directors or advisory committees for Novartis, Takeda, Roche, and Celgene, and received research funding from Roche. P.B. provided consultant services to and received honoraria from Novartis. C.d.C., L.P., and V.V.R. were employed by Novartis. R.T. was employed by Novartis Healthcare Private Limited. Ö.A. was employed by Novartis Pharma AG. R.A. was employed by Novartis Institutes for BioMedical Research, and holds equity ownership in Novartis, Cara Therapeutics, Aeterna Zentaris, Exelixis, and Celgene. S.J.S. provided consultant services to, received honoraria from, was a member of an entity’s board of directors or advisory committees for, and received research funding from Celgene; provided consultant services to and received honoraria from Dava Oncology; received honoraria and research funding from Genentech; was a member of an entity’s board of directors or advisory committees for Gilead and Pfizer; provided consultant services to, received honoraria from, and received research funding from Merck; received honoraria from, was a member of an entity’s board of directors or advisory committees for and received research funding from Novartis; provided consultant services to, received honoraria from, and was a member of an entity’s board of directors or advisory committees for Nordic Nanovector; and received honoraria from OncLive and Physician’s Education Source.
Figures

References
-
- Van Den Neste E, Schmitz N, Mounier N, et al. Outcome of patients with relapsed diffuse large B-cell lymphoma who fail second-line salvage regimens in the International CORAL study. Bone Marrow Transplant. 2016;51(1):51-57. - PubMed
-
- Gisselbrecht C, Schmitz N, Mounier N, et al. Rituximab maintenance therapy after autologous stem-cell transplantation in patients with relapsed CD20(+) diffuse large B-cell lymphoma: final analysis of the collaborative trial in relapsed aggressive lymphoma. J Clin Oncol. 2012;30(36):4462-4469. - PMC - PubMed
-
- Schuster SJ, Bishop MR, Tam CS, et al. ; JULIET Investigators . Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380(1):45-56. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

