From Synthetic Fragments of Endogenous Three-Finger Proteins to Potential Drugs
- PMID: 31333465
- PMCID: PMC6616073
- DOI: 10.3389/fphar.2019.00748
From Synthetic Fragments of Endogenous Three-Finger Proteins to Potential Drugs
Abstract
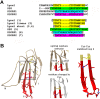
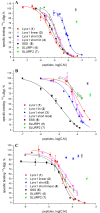
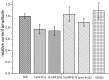
The proteins of the Ly6 family have a three-finger folding as snake venom α-neurotoxins, targeting nicotinic acetylcholine receptors (nAChRs), and some of them, like mammalian secreted Ly6/uPAR protein (SLURP1) and membrane-attached Ly-6/neurotoxin (Lynx1), also interact with distinct nAChR subtypes. We believed that synthetic fragments of these endogenous proteins might open new ways for drug design because nAChRs are well-known targets for developing analgesics and drugs against neurodegenerative diseases. Since interaction with nAChRs was earlier shown for synthetic fragments of the α-neurotoxin central loop II, we synthesized a 15-membered fragment of human Lynx1, its form with two Cys residues added at the N- and C-termini and forming a disulfide, as well as similar forms of human SLURP1, SLURP2, and of Drosophila sleepless protein (SSS). The IC50 values measured in competition with radioiodinated α-bungarotoxin for binding to the membrane-bound Torpedo californica nAChR were 4.9 and 7.4 µM for Lynx1 and SSS fragments, but over 300 µM for SLURP1 or SLURP2 fragments. The affinity of these compounds for the α7 nAChR in the rat pituitary tumor-derived cell line GH4C1 was different: 13.1 and 147 µM for SSS and Lynx1 fragments, respectively. In competition for the ligand-binding domain of the α9 nAChR subunit, SSS and Lynx1 fragments had IC50 values of about 40 µM, which correlates with the value found for the latter with the rat α9α10 nAChR expressed in the Xenopus oocytes. Thus, the activity of these synthetic peptides against muscle-type and α9α10 nAChRs indicates that they may be useful in design of novel myorelaxants and analgesics.
Keywords: Ly6 family; nAChRs; nicotinic acetylcholine receptors; peptide fragments; three-finger proteins.
Figures

References
LinkOut - more resources
Full Text Sources

