Vascular and neural basis of the BOLD signal
- PMID: 31336326
- PMCID: PMC6859204
- DOI: 10.1016/j.conb.2019.06.004
Vascular and neural basis of the BOLD signal
Abstract
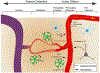
Neural activity in the brain is usually coupled to increases in local cerebral blood flow, leading to the increase in oxygenation that generates the BOLD fMRI signal. Recent work has begun to elucidate the vascular and neural mechanisms underlying the BOLD signal. The dilatory response is distributed throughout the vascular network. Arteries actively dilate within a second following neural activity increases, while venous distensions are passive and have a time course that last tens of seconds. Vasodilation, and thus local blood flow, is controlled by the activity of both neurons and astrocytes via multiple different pathways. The relationship between sensory-driven neural activity and the vascular dynamics in sensory areas are well-captured with a linear convolution model. However, depending on the behavioral state or brain region, the coupling between neural activity and hemodynamic signals can be weak or even inverted.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Figures


References
-
-
Drew PJ, Shih AY, Kleinfeld D: Fluctuating and sensory-induced vasodynamics in rodent cortex extend arteriole capacity. Proc Natl Acad Sci USA 2011, 108:8473–8478.
* The first two-photon measurements of arterial and venous dilations in the awake animal.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

