Dihydrogen Bond in the Aminoborane Complex of a Nicergoline Intermediate
- PMID: 31336918
- PMCID: PMC6680414
- DOI: 10.3390/molecules24142548
Dihydrogen Bond in the Aminoborane Complex of a Nicergoline Intermediate
Abstract
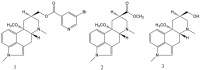
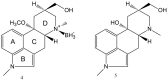
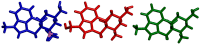
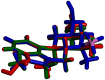
An aminoborane side product from the nicergoline manufacture process was identified by single-crystal X-ray diffraction. As boranes of pharmaceutical molecules are quite rare, the binding potential of the BH3 group was investigated and compared with similar compounds using Cambridge Structural Database (CSD). Surprisingly, the packing was stabilized by a dihydrogen bond, which triggered a false alert for too-short contact of hydrogen atoms in IUCR checkCIF. As the dihydrogen bond concept is not widely known, such an alert might mislead crystallographers to force -CH3 optimal geometry to -BH3 groups. The B-H distances equal to or less than 1.0 Å (17% of the CSD structures) are substantially biased when analyzing the structures of aminoborane complexes in CSD. To conduct proper searching, B-H bond length normalization should be applied in the CSD search.
Keywords: CSD; X-ray structure; aminoborane; dihydrogen bond; nicergoline; single crystal.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


References
-
- Schweitzer B.A., Egholm M., Koch T.H. Mechanistic studies of the reduction of daunomycin with sodium borohydride. Formation and reaction of borate esters. J. Am. Chem. Soc. 1992;114:242–248. doi: 10.1021/ja00027a031. - DOI
-
- Liu Q., Xiong F.-J., He Q.-Q., Chen F.-E. Development of an Efficient Process for the Decomposition of the Borate Complexes Formed during the Large-Scale Synthesis of (S)-1,2,4-Butanetriol. Org. Process Res. Dev. 2013;17:1540–1542. doi: 10.1021/op400271k. - DOI
-
- Niedenzu K., Dawson J.W. Boron-Nitrogen Compounds. III.1,2 Aminoboranes, Part 2: The B-N Bond Character in Substituted Aminoboranes. J. Am. Chem. Soc. 1960;82:4223–4228. doi: 10.1021/ja01501a028. - DOI
-
- Narayana C., Periasamy M. Hydroboration of prochiral olefins with chiral Lewis base–borane complexes: Relationship to the mechanism of hydroboration. J. Chem. Soc. Chem. Commun. 1987:1857–1859. doi: 10.1039/C39870001857. - DOI
-
- Le Toumelin J.-B., Baboulène M. Chiral intramolecular amine-borane complexes as reducing agents for prochiral ketones. Tetrahedron Asymmetry. 1997;8:1259–1265. doi: 10.1016/S0957-4166(97)00093-1. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

