Docosahexaenoic Acid Enhances Oxaliplatin-Induced Autophagic Cell Death via the ER Stress/Sesn2 Pathway in Colorectal Cancer
- PMID: 31337142
- PMCID: PMC6678695
- DOI: 10.3390/cancers11070982
Docosahexaenoic Acid Enhances Oxaliplatin-Induced Autophagic Cell Death via the ER Stress/Sesn2 Pathway in Colorectal Cancer
Abstract
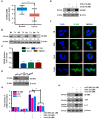
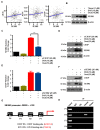
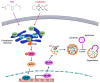
Oxaliplatin is an anticancer drug administered to colorectal cancer (CRC) patients in combination with 5-fluorouracil and antibodies (bevacizumab and cetuximab), thereby significantly improving the survival rate of CRC. However, due to various side effects associated with the above treatment strategy, the need for combinatorial therapeutic strategies has emerged. Based on the demand for new combinatorial therapies and the known antitumor effects of the omega-3 polyunsaturated fatty acid, docosahexaenoic acid (DHA), we investigated the Oxaliplatin and DHA combination for its effect. Our results indicated that DHA further enhanced Oxaliplatin-induced cell viability and autophagic cell death, in vitro and in vivo. Oxaliplatin and DHA also increased the expression of Sestrin 2 (SESN2) and endoplasmic reticulum (ER) stress related C/EBP homologous protein (CHOP). Additionally, treatment with Oxaliplatin and DHA enhanced the binding of CHOP to the promotor region of SESN2, increasing SESN2 expression. These results suggested that DHA enhanced Oxaliplatin-induced reduction in cell viability and increase in autophagy via activating SESN2 and increasing ER stress. Thus, SESN2 may be an effective preclinical target for CRC treatment.
Keywords: Oxaliplatin; Sestrin 2; autophagic cell death; colon cancer; docosahexaenoic acid.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




References
-
- Bendell J.C., Bekaii-Saab T.S., Cohn A.L., Hurwitz H.I., Kozloff M., Tezcan H., Roach N., Mun Y., Fish S., Flick E.D., et al. Treatment patterns and clinical outcomes in patients with metastatic colorectal cancer initially treated with FOLFOX-bevacizumab or FOLFIRI-bevacizumab: Results from ARIES, a bevacizumab observational cohort study. Oncologist. 2012;17:1486–1495. doi: 10.1634/theoncologist.2012-0190. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

