Clinical and Molecular Predictors of Response to Immune Checkpoint Inhibitors in Patients with Advanced Esophagogastric Cancer
- PMID: 31337644
- PMCID: PMC6905384
- DOI: 10.1158/1078-0432.CCR-18-3603
Clinical and Molecular Predictors of Response to Immune Checkpoint Inhibitors in Patients with Advanced Esophagogastric Cancer
Abstract
Purpose: Immune checkpoint inhibitors (ICI) are effective in only a minority of patients with esophagogastric cancer (EGC). Here, we aimed to identify predictors of durable clinical benefit to ICI in EGC.
Experimental design: Patients with advanced EGC treated with ICIs at Memorial Sloan Kettering Cancer Center (New York, NY) were identified. Clinicopathologic variables were assessed. In patients profiled by MSK-IMPACT-targeted sequencing, outcomes were correlated with tumor genomic features.
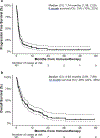
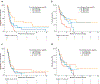
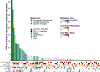
Results: One-hundred sixty-one patients were treated with ICIs (110 with anti-PD-1/PD-L1 antibodies and 51 with anti-CTLA-4 and PD-1/PD-L1 antibodies). The median progression-free survival (PFS) and overall survival (OS) were 1.7 and 4.9 months, respectively. Greater number of disease sites (≥3), liver metastases, treatment with ≥3 prior therapies and ECOG performance status ≥2 were associated with poorer PFS and OS. Patients treated with combination ICI and those with PD-L1-positive tumors had improved outcomes. There was no difference in outcomes between patients treated with antibiotics during or in the 2 months preceding ICI treatment versus those who were not. Occurrence of irAEs was associated with improved OS. In genomically profiled tumors (n = 89), survival was associated with increasing tumor mutation burden (TMB). However, in multivariable analyses and when microsatellite unstable (MSI) patients were excluded, a significant association was no longer observed.
Conclusions: In patients with advanced EGC, heavily pretreated patients, those with high-volume disease and/or poor PS were less likely to benefit from ICI. irAEs were associated with improved OS. TMB correlated with improved survival, but this association was not observed when MSI-high patients were excluded.
©2019 American Association for Cancer Research.
Figures
Similar articles
-
The efficacy and safety of PD-1/PD-L1 inhibitors in combination with chemotherapy as a first-line treatment for unresectable, locally advanced, HER2-negative gastric or gastroesophageal junction cancer: a meta-analysis of randomized controlled trials.Front Immunol. 2025 Mar 26;16:1566939. doi: 10.3389/fimmu.2025.1566939. eCollection 2025. Front Immunol. 2025. PMID: 40207218 Free PMC article.
-
Spatial and Temporal Heterogeneity of PD-L1 Expression and Tumor Mutational Burden in Gastroesophageal Adenocarcinoma at Baseline Diagnosis and after Chemotherapy.Clin Cancer Res. 2020 Dec 15;26(24):6453-6463. doi: 10.1158/1078-0432.CCR-20-2085. Epub 2020 Aug 20. Clin Cancer Res. 2020. PMID: 32820017 Free PMC article.
-
Safety and efficacy of pembrolizumab in combination with S-1 plus oxaliplatin as a first-line treatment in patients with advanced gastric/gastroesophageal junction cancer: Cohort 1 data from the KEYNOTE-659 phase IIb study.Eur J Cancer. 2020 Apr;129:97-106. doi: 10.1016/j.ejca.2020.02.002. Epub 2020 Mar 4. Eur J Cancer. 2020. PMID: 32145474 Clinical Trial.
-
Analysis of the treatment efficacy and prognostic factors of PD-1/PD-L1 inhibitors for advanced gastric or gastroesophageal junction cancer: a multicenter, retrospective clinical study.Front Immunol. 2024 Oct 24;15:1468342. doi: 10.3389/fimmu.2024.1468342. eCollection 2024. Front Immunol. 2024. PMID: 39512347 Free PMC article.
-
PD-1 and PD-L1 inhibitors in oesophago-gastric cancers.Cancer Lett. 2020 Jan 28;469:142-150. doi: 10.1016/j.canlet.2019.10.036. Epub 2019 Oct 25. Cancer Lett. 2020. PMID: 31669518 Review.
Cited by
-
Prediction of immune checkpoint inhibition with immune oncology-related gene expression in gastrointestinal cancer using a machine learning classifier.J Immunother Cancer. 2020 Aug;8(2):e000631. doi: 10.1136/jitc-2020-000631. J Immunother Cancer. 2020. PMID: 32792359 Free PMC article.
-
AFF3 is a novel prognostic biomarker and a potential target for immunotherapy in gastric cancer.J Clin Lab Anal. 2022 Jun;36(6):e24437. doi: 10.1002/jcla.24437. Epub 2022 Apr 27. J Clin Lab Anal. 2022. PMID: 35478418 Free PMC article.
-
PD-1 inhibitors versus chemotherapy as second-line treatment for advanced esophageal squamous cell carcinoma: a meta-analysis.BMC Cancer. 2021 Nov 10;21(1):1195. doi: 10.1186/s12885-021-08958-3. BMC Cancer. 2021. PMID: 34758782 Free PMC article.
-
The impact of antibiotic use on clinical features and survival outcomes of cancer patients treated with immune checkpoint inhibitors.Front Immunol. 2022 Jul 28;13:968729. doi: 10.3389/fimmu.2022.968729. eCollection 2022. Front Immunol. 2022. PMID: 35967438 Free PMC article.
-
Delineating Molecular Subtypes through Gene Set Variation Analysis Confers Therapeutic and Prognostic Capability in Gastric Cancer.Can J Gastroenterol Hepatol. 2022 Jul 15;2022:5415758. doi: 10.1155/2022/5415758. eCollection 2022. Can J Gastroenterol Hepatol. 2022. PMID: 35875363 Free PMC article.
References
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A: Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin, 2018 - PubMed
-
- Kang YK, Boku N, Satoh T, Ryu MH, Chao Y, Kato K, et al.: Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538–12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet, 2017 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

