Neurodevelopmental Outcomes of Preterm Infants With Retinopathy of Prematurity by Treatment
- PMID: 31337693
- PMCID: PMC6855825
- DOI: 10.1542/peds.2018-3537
Neurodevelopmental Outcomes of Preterm Infants With Retinopathy of Prematurity by Treatment
Abstract
Objective: Among extremely preterm infants, we evaluated whether bevacizumab therapy compared with surgery for retinopathy of prematurity (ROP) is associated with adverse outcomes in early childhood.
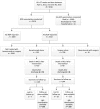
Methods: This study was a retrospective analysis of prospectively collected data on preterm (22-26 + 6/7 weeks' gestational age) infants admitted to the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network centers who received bevacizumab or surgery exclusively for ROP. The primary outcome was death or severe neurodevelopmental impairment (NDI) at 18 to 26 months' corrected age (Bayley Scales of Infant and Toddler Development, Third Edition cognitive or motor composite score <70, Gross Motor Functional Classification Scale level ≥2, bilateral blindness or hearing impairment).
Results: The cohort (N = 405; 214 [53%] boys; median [interquartile range] gestational age: 24.6 [23.9-25.3] weeks) included 181 (45%) infants who received bevacizumab and 224 (55%) who underwent ROP surgery. Infants treated with bevacizumab had a lower median (interquartile range) birth weight (640 [541-709] vs 660 [572.5-750] g; P = .02) and longer durations of conventional ventilation (35 [21-58] vs 33 [18-49] days; P = .04) and supplemental oxygen (112 [94-120] vs 105 [84.5-120] days; P = .01). Death or severe NDI (adjusted odds ratio [aOR] 1.42; 95% confidence interval [CI] 0.94 to 2.14) and severe NDI (aOR 1.14; 95% CI 0.76 to 1.70) did not differ between groups. Odds of death (aOR 2.54 [95% CI 1.42 to 4.55]; P = .002), a cognitive score <85 (aOR 1.78 [95% CI 1.09 to 2.91]; P = .02), and a Gross Motor Functional Classification Scale level ≥2 (aOR 1.73 [95% CI 1.04 to 2.88]; P = .04) were significantly higher with bevacizumab therapy.
Conclusions: In this multicenter cohort of preterm infants, ROP treatment modality was not associated with differences in death or NDI, but the bevacizumab group had higher mortality and poor cognitive outcomes in early childhood. These data reveal the need for a rigorous appraisal of ROP therapy.
Copyright © 2019 by the American Academy of Pediatrics.
Conflict of interest statement
POTENTIAL CONFLICT OF INTEREST: The authors have indicated they have no potential conflicts of interest to disclose.
Figures
References
-
- Mintz-Hittner HA. Treatment of retinopathy of prematurity with vascular endothelial growth factor inhibitors. Early Hum Dev. 2012;88(12):937–941 - PubMed
-
- Cernichiaro-Espinosa LA, Olguin-Manriquez FJ, Henaine-Berra A, Garcia-Aguirre G, Quiroz-Mercado H, Martinez-Castellanos MA. New insights in diagnosis and treatment for retinopathy of prematurity. Int Ophthalmol. 2016;36(5):751–760 - PubMed
-
- Lepore D, Quinn GE, Molle F, et al. Intravitreal bevacizumab versus laser treatment in type 1 retinopathy of prematurity: report on fluorescein angiographic findings. Ophthalmology. 2014;121(11):2212–2219 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- UG1 HD068263/HD/NICHD NIH HHS/United States
- U10 HD021373/HD/NICHD NIH HHS/United States
- UL1 TR001111/TR/NCATS NIH HHS/United States
- K23 DK120960/DK/NIDDK NIH HHS/United States
- UG1 HD068270/HD/NICHD NIH HHS/United States
- U10 HD040689/HD/NICHD NIH HHS/United States
- UG1 HD027856/HD/NICHD NIH HHS/United States
- UG1 HD034216/HD/NICHD NIH HHS/United States
- UG1 HD027904/HD/NICHD NIH HHS/United States
- UG1 HD027880/HD/NICHD NIH HHS/United States
- UL1 TR001117/TR/NCATS NIH HHS/United States
- UG1 HD027851/HD/NICHD NIH HHS/United States
- U10 HD036790/HD/NICHD NIH HHS/United States
- UG1 HD068278/HD/NICHD NIH HHS/United States
- UG1 HD068244/HD/NICHD NIH HHS/United States
- UG1 HD027853/HD/NICHD NIH HHS/United States
- UG1 HD087229/HD/NICHD NIH HHS/United States
- UG1 HD040689/HD/NICHD NIH HHS/United States
- M01 RR008084/RR/NCRR NIH HHS/United States
- UG1 HD053089/HD/NICHD NIH HHS/United States
- UG1 HD068284/HD/NICHD NIH HHS/United States
- UG1 HD021385/HD/NICHD NIH HHS/United States
- UG1 HD040492/HD/NICHD NIH HHS/United States
- UG1 HD021364/HD/NICHD NIH HHS/United States
- UG1 HD053109/HD/NICHD NIH HHS/United States
- U10 HD068278/HD/NICHD NIH HHS/United States