Use of Body Surface Area for Dosing of Vancomycin
- PMID: 31337992
- PMCID: PMC6633278
- DOI: 10.5863/1551-6776-24.4.296
Use of Body Surface Area for Dosing of Vancomycin
Abstract
Objectives: Vancomycin weight-based dosing regimens often fail to achieve therapeutic trough serum concentration in children ≤12 years of age and rigorous studies evaluating efficacy and safety of body surface area (BSA)-based dosing regimens have not been performed. We compared vancomycin trough serum concentrations in pediatric patients receiving a weight- or BSA-based dosing regimen.
Methods: This was a single-center, retrospective study evaluating pediatric patients, ages 1 to 12 years, who received vancomycin from September 2012 to October 2015. Patients received a minimum of 3 consecutive doses at the same scheduled interval within a dosing regimen prior to a measured vancomycin serum trough concentration. The primary outcome was percentage of initial vancomycin trough concentrations ≥10 mg/L. The secondary outcomes were percentage of supratherapeutic, therapeutic, and subtherapeutic vancomycin serum concentration for all patients, including a subset of overweight and obese patients, and number of nephrotoxic occurrences.
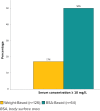
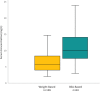
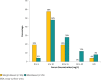
Results: BSA-based dosing regimens resulted in 50% of the initial vancomycin trough concentrations ≥ 10 mg/L compared with 17% for the weight-based dosing regimens (p < 0.0001). No statistically significant differences were noted between the 2 dosing regimens for supratherapeutic, therapeutic, or subtherapeutic trough concentrations for all patients, and for the subset of overweight and obese patients. Nephrotoxic occurrences were noted in 7% of the weight-based dosing regimens compared with none in the BSA-based dosing regimens.
Conclusions: A BSA-based vancomycin dosing regimen resulted in significantly more initial vancomycin trough concentrations ≥10 mg/L and trended towards higher initial vancomycin trough concentrations without observable nephrotoxicity.
Keywords: antibiotic dosing; body surface area; pediatrics; therapeutic drug monitoring; vancomycin.
Conflict of interest statement
Disclosure The authors declare no conflicts or financial interest in any product or service mentioned in the manuscript, including grants, equipment, medications, employment, gifts, and honoraria. Beth Sawrey and Mary Subramanian have full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Figures

References
-
- Dehority W. Use of vancomycin in pediatrics. Pediatr Infect Dis J. 2010;29(5):462–464. - PubMed
-
- Liu C, Bayer A, Cosgrove SE et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):1–38. - PubMed
-
- McNeil JC, Kaplan SL, Vallego JG. The influence of the route of antibiotic administration, methicillin susceptibility, vancomycin duration and serum trough concentration on outcomes of pediatric Staphylococcus aureus bacteremic osteoarticular infection. Pediatric Infec Dis J. 2017;36(6):572–577. - PMC - PubMed
LinkOut - more resources
Full Text Sources
